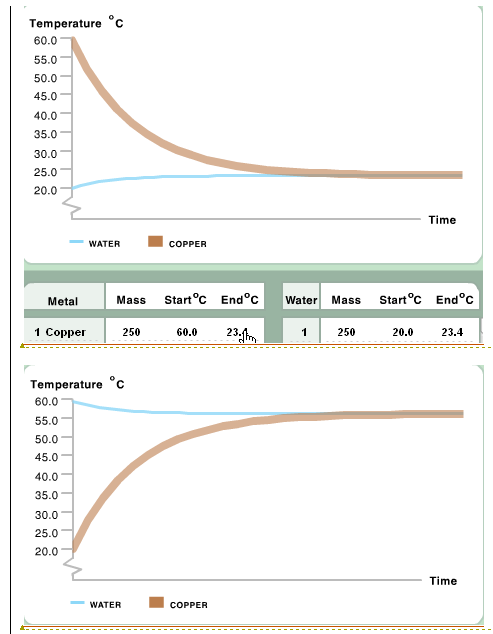
Specific heat capacity Physics Homework Help, Physics Assignments and Projects Help, Assignments Tutors online

SOLVED: Question 1 (1 point) Given the following specific value data, which statement is correct? Metal lead bronze Cue'c) 028 brass coppel nickel cobalt stee aluminum chromium Cnnttuum 0.23 Given the same
Why isn't the specific heat capacity of brass the average of the specific heat capacity of its components (which are copper and zinc)? Both copper and zinc have values of 376.8 J/(kg*K)

How to determine specific heat capacity of materials thermal energy store uses examples applications igcse/O level/gcse Physics revision notes