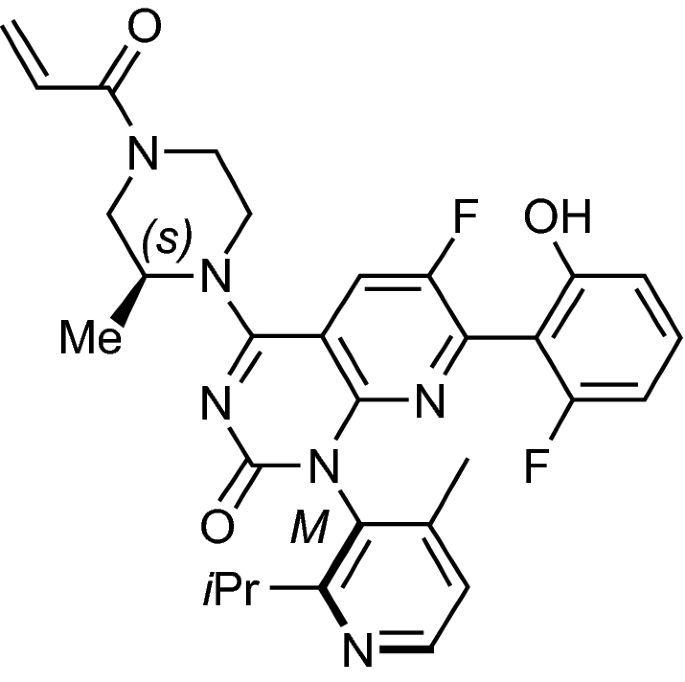
Similarities and differences among sotorasib and adagrasib (direct KRAS... | Download Scientific Diagram
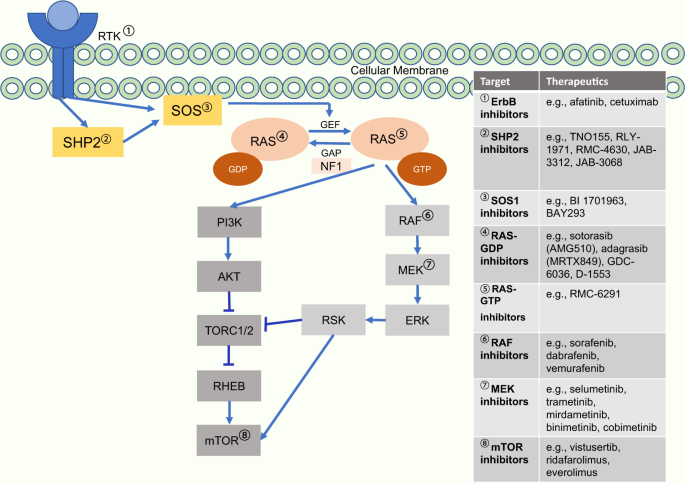
Selective KRAS G12C inhibitors in non-small cell lung cancer: chemistry, concurrent pathway alterations, and clinical outcomes | npj Precision Oncology

Sotorasib for previously treated colorectal cancers with KRASG12C mutation (CodeBreaK100): a prespecified analysis of a single-arm, phase 2 trial - The Lancet Oncology

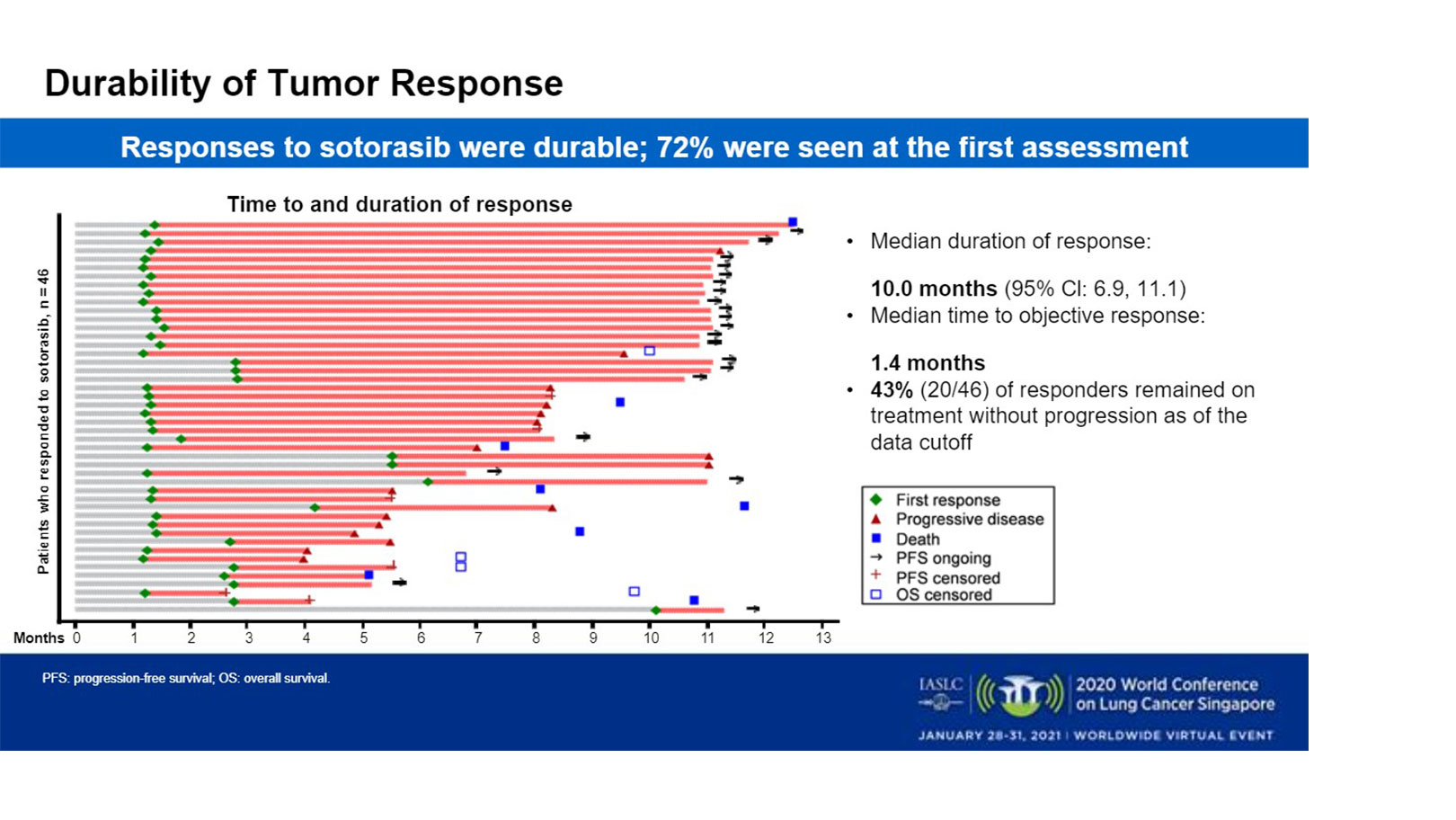
Stephen V Liu, MD on Twitter: "#ESMO20 Overall, sotorasib showing clear efficacy (RR 32.2%, DOR 10.9m, PFS 6.3m) and excellent safety profile. Would have a clear role today in salvage setting. Good
![Absorption, metabolism and excretion of [14C]-sotorasib in healthy male subjects: characterization of metabolites and a minor albumin-sotorasib conjugate | springermedizin.de Absorption, metabolism and excretion of [14C]-sotorasib in healthy male subjects: characterization of metabolites and a minor albumin-sotorasib conjugate | springermedizin.de](https://media.springernature.com/lw150/springer-static/cover/journal/280/90/4.jpg?as=jpg)
Absorption, metabolism and excretion of [14C]-sotorasib in healthy male subjects: characterization of metabolites and a minor albumin-sotorasib conjugate | springermedizin.de
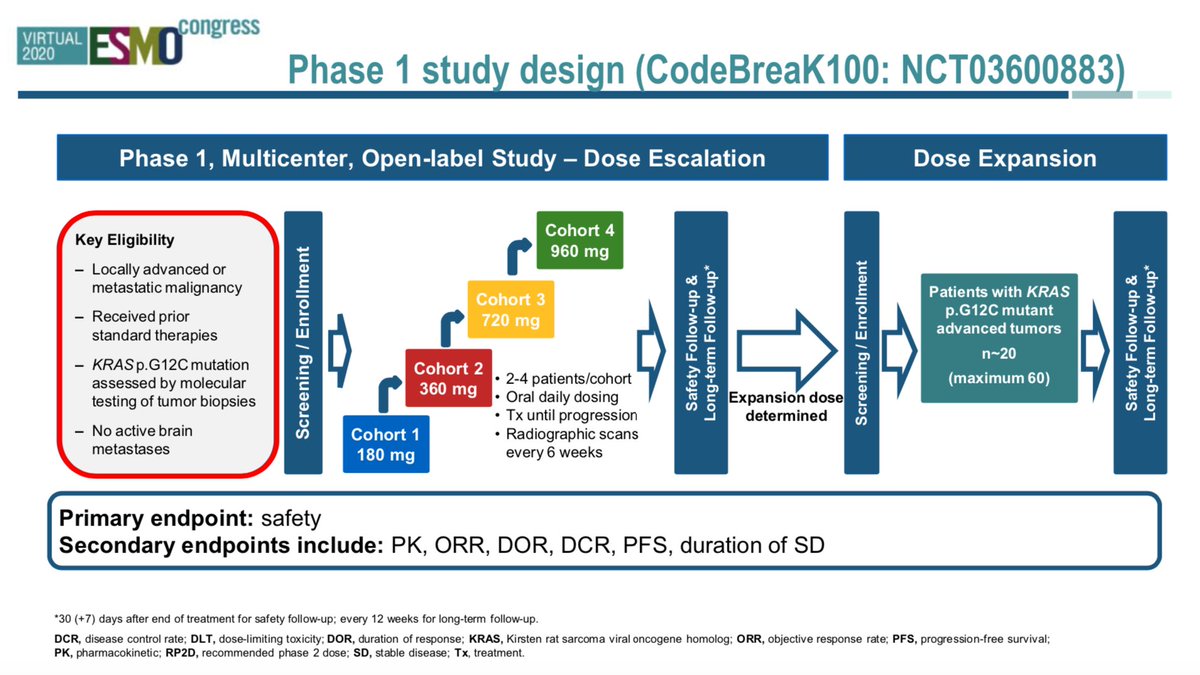
Stephen V Liu, MD on Twitter: "#ESMO20 CodeBreak100 is a phase I escalation/expansion trial of sotorasib monotherapy in KRAS G12C tumors. Primary endpoint was safety. Escalation established preferred dose of 960mg qday. #

KRAS inhibitor sotorasib appears safe, achieves durable clinical benefit in early trial | MD Anderson Cancer Center
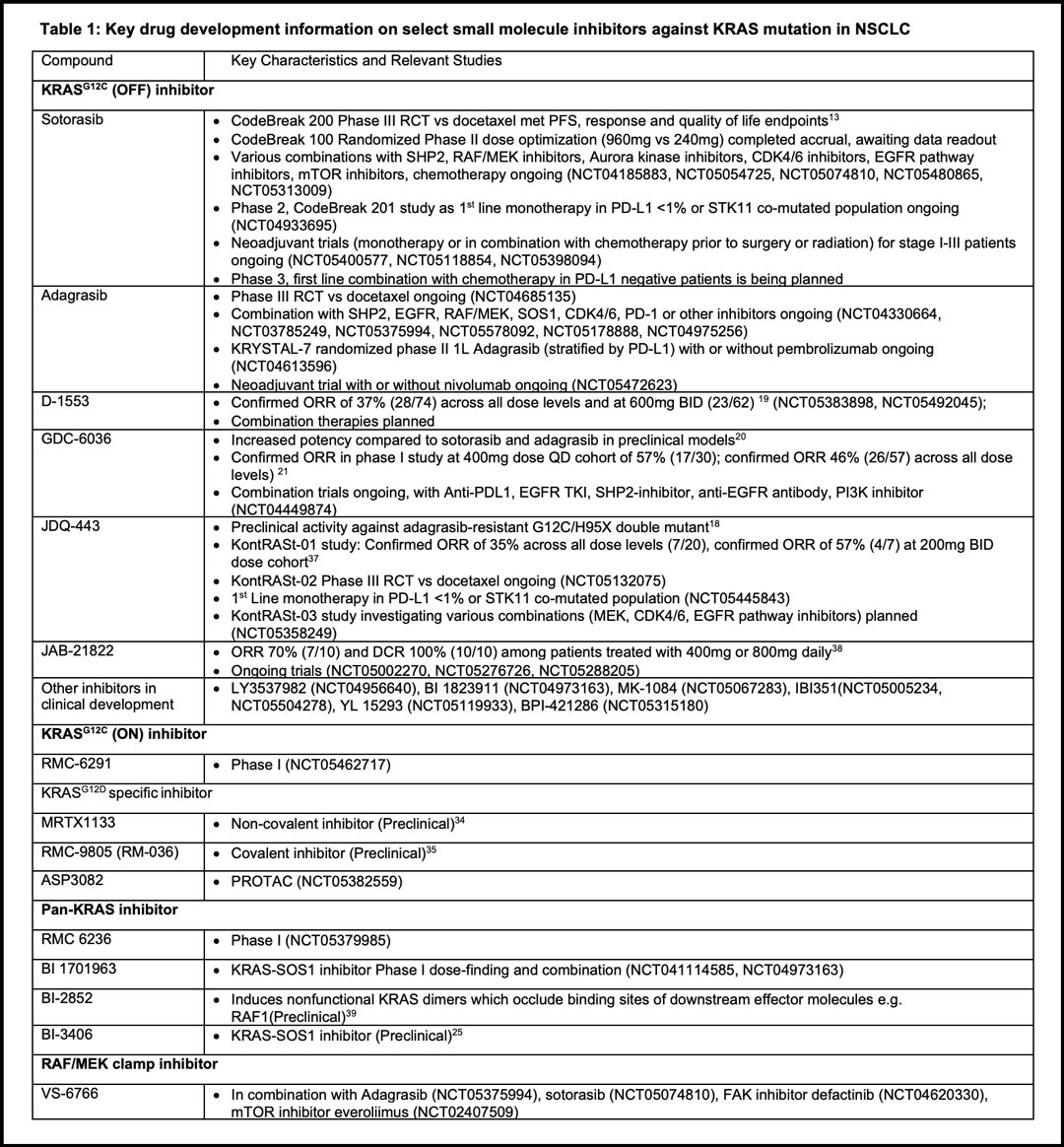
Undruggable No More: KRAS Experts Review What is Known—and Unknown—About This Challenging Alteration - ILCN.org (ILCN/WCLC)
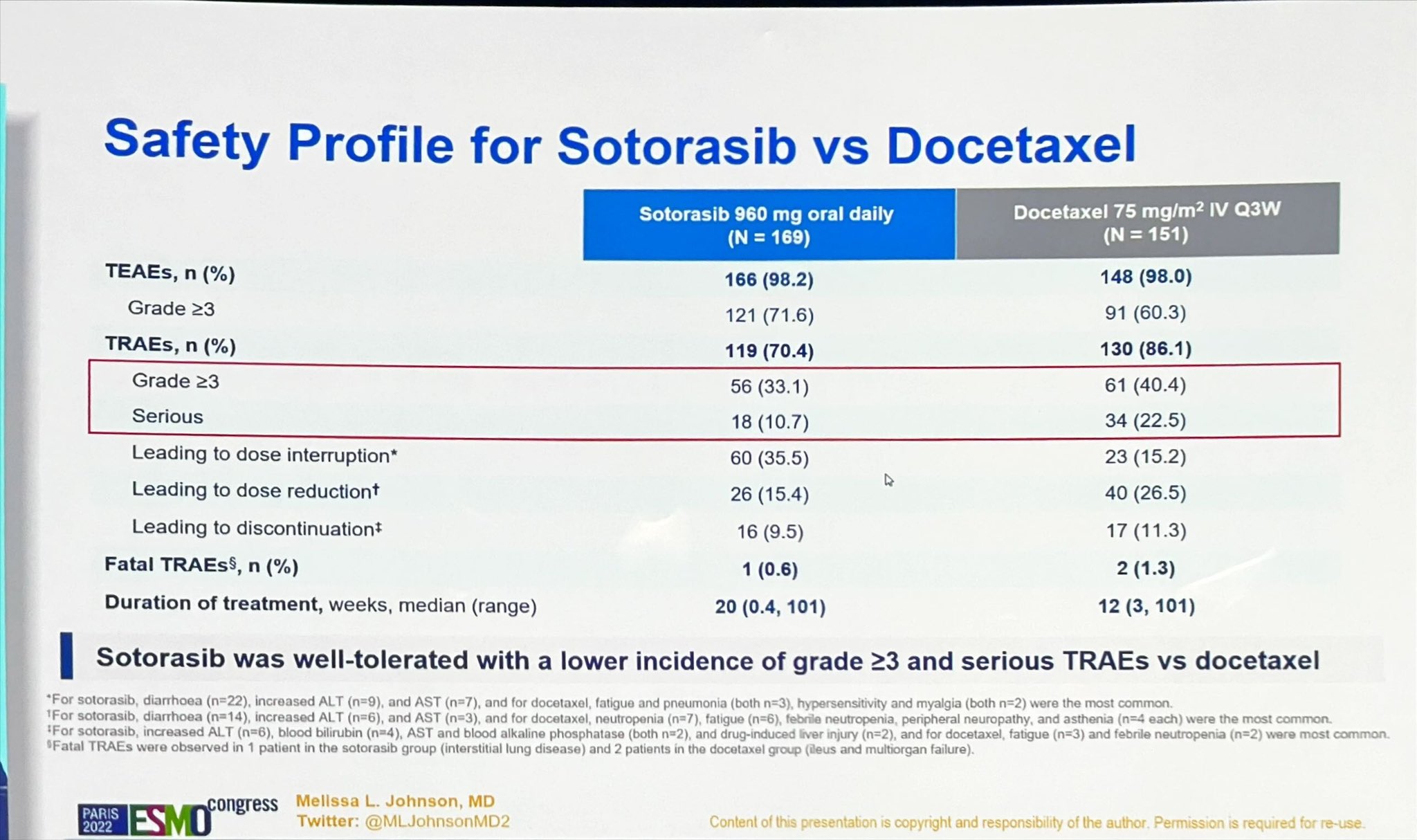
Dr. Antonio Calles 🫁🚭 on Twitter: "💥 Sotorasib vs docetaxel for previously treated NSCLC with KRAS G12C mutation: CodeBreak200 phase IlI study 😕 ORR 28.1% 😕 PFS 5.6 months (delta 1.1 month)