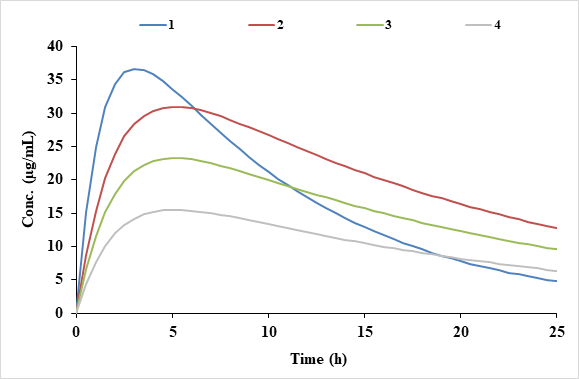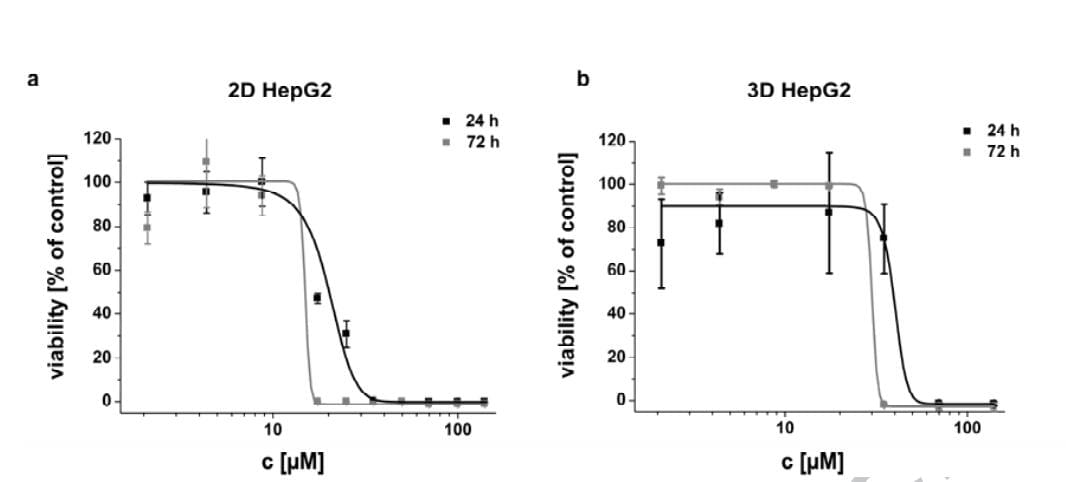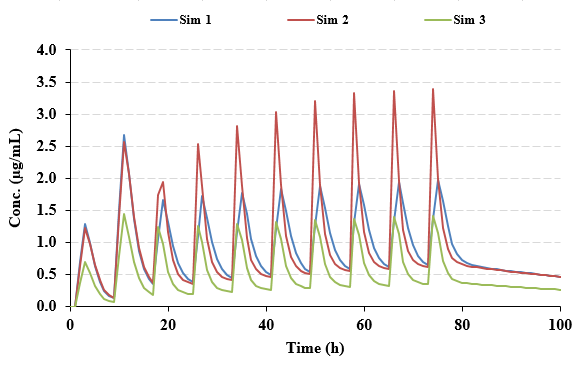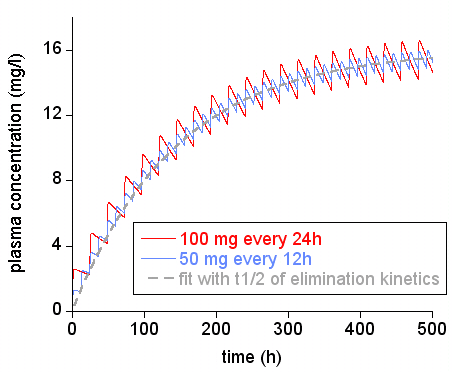
Functional assessments in repeat-dose toxicity studies: the art of the possible - Toxicology Research (RSC Publishing)
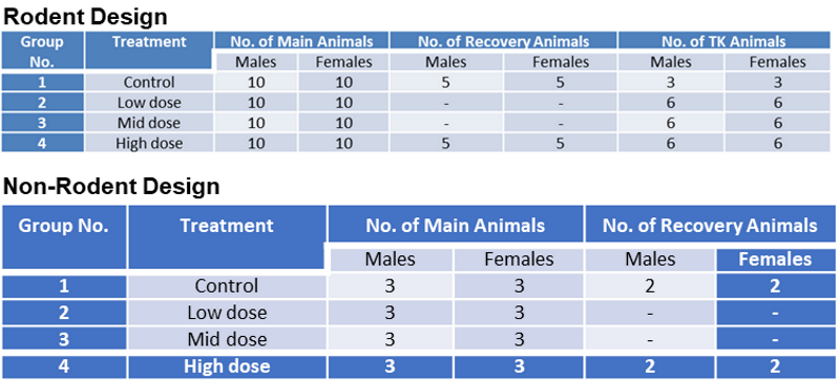
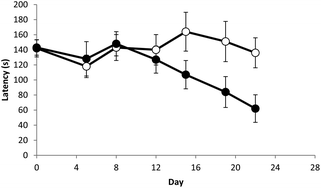
Assessment of acute, 14-day, and 13-week repeated oral dose toxicity of Tiglium seed extract in rats | BMC Complementary Medicine and Therapies | Full Text

Toxicology 90 Days Long Terms Study Repeated Dose Intramuscular Toxicity Study/IC, Pre Clinical Studies, Rs 200000/test | ID: 6249089462
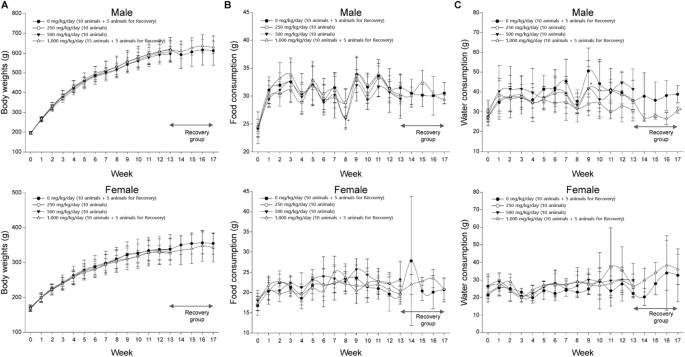
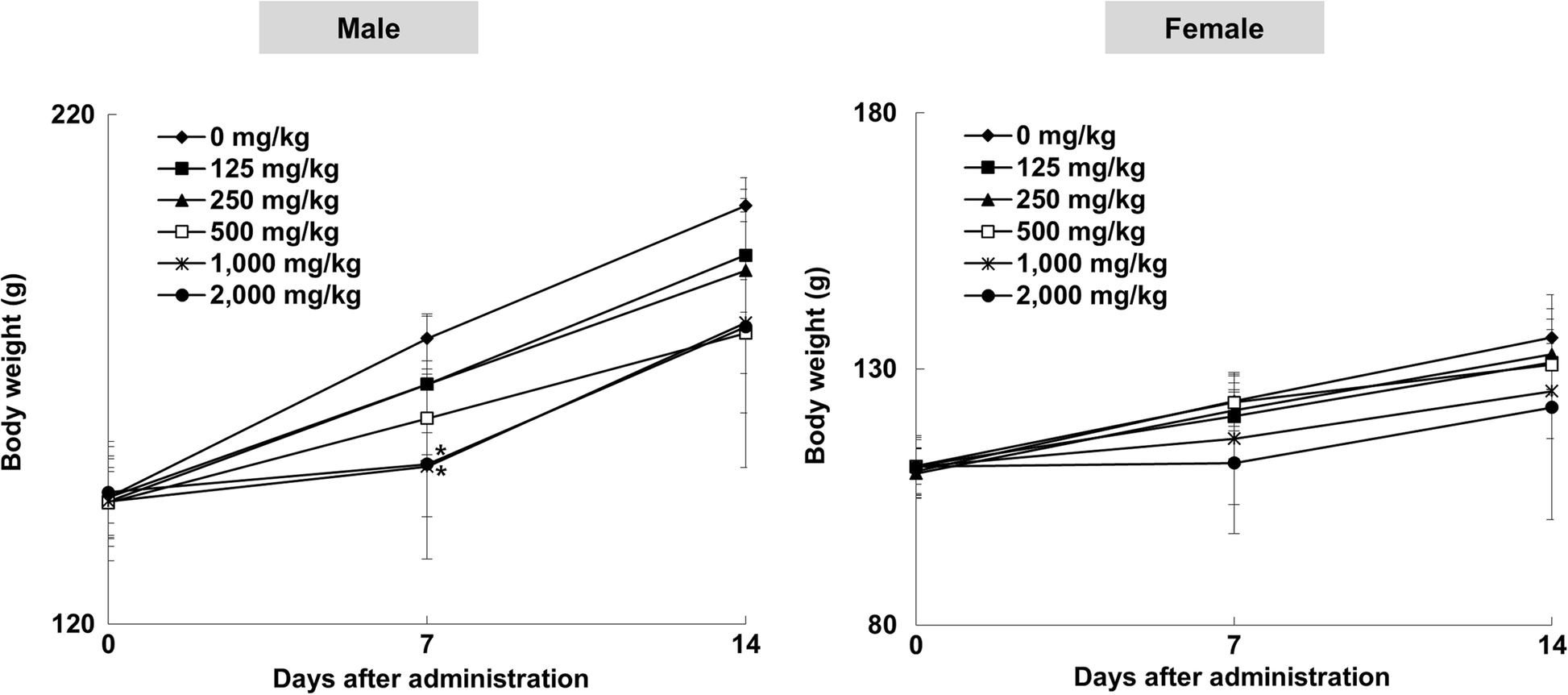
Oral toxicity of titanium dioxide P25 at repeated dose 28-day and 90-day in rats | Particle and Fibre Toxicology | Full Text

Repeat Dosing of AAV2.5T to Ferret Lungs Elicits an Antibody Response That Diminishes Transduction in an Age-Dependent Manner: Molecular Therapy - Methods & Clinical Development

Scheme of repeated dose toxicity study for VGX-6150. C57BL/6 mice were... | Download Scientific Diagram
![PDF] 28 day repeat dose (sub-chronic) toxicity of recombinant Enfuviritide on New zealand white rabbits | Semantic Scholar PDF] 28 day repeat dose (sub-chronic) toxicity of recombinant Enfuviritide on New zealand white rabbits | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/5b8fc7d91088195b2fe86e31521777ff3dafe9d3/4-TableII-1.png)
PDF] 28 day repeat dose (sub-chronic) toxicity of recombinant Enfuviritide on New zealand white rabbits | Semantic Scholar

Repeat dose evinacumab in patients with severe hypertriglyceridaemia (HTG) at risk of pancreatitis (NCT03452228) - Triglyceride Forum
Recommendations on dose level selection for repeat dose toxicity studies,Archives of Toxicology - X-MOL

First study of repeat dose co-administration of GSK1265744 and TMC278 long-acting parenteral nanosuspensions: pharmacokinetics, safety, and tolerability in healthy adults