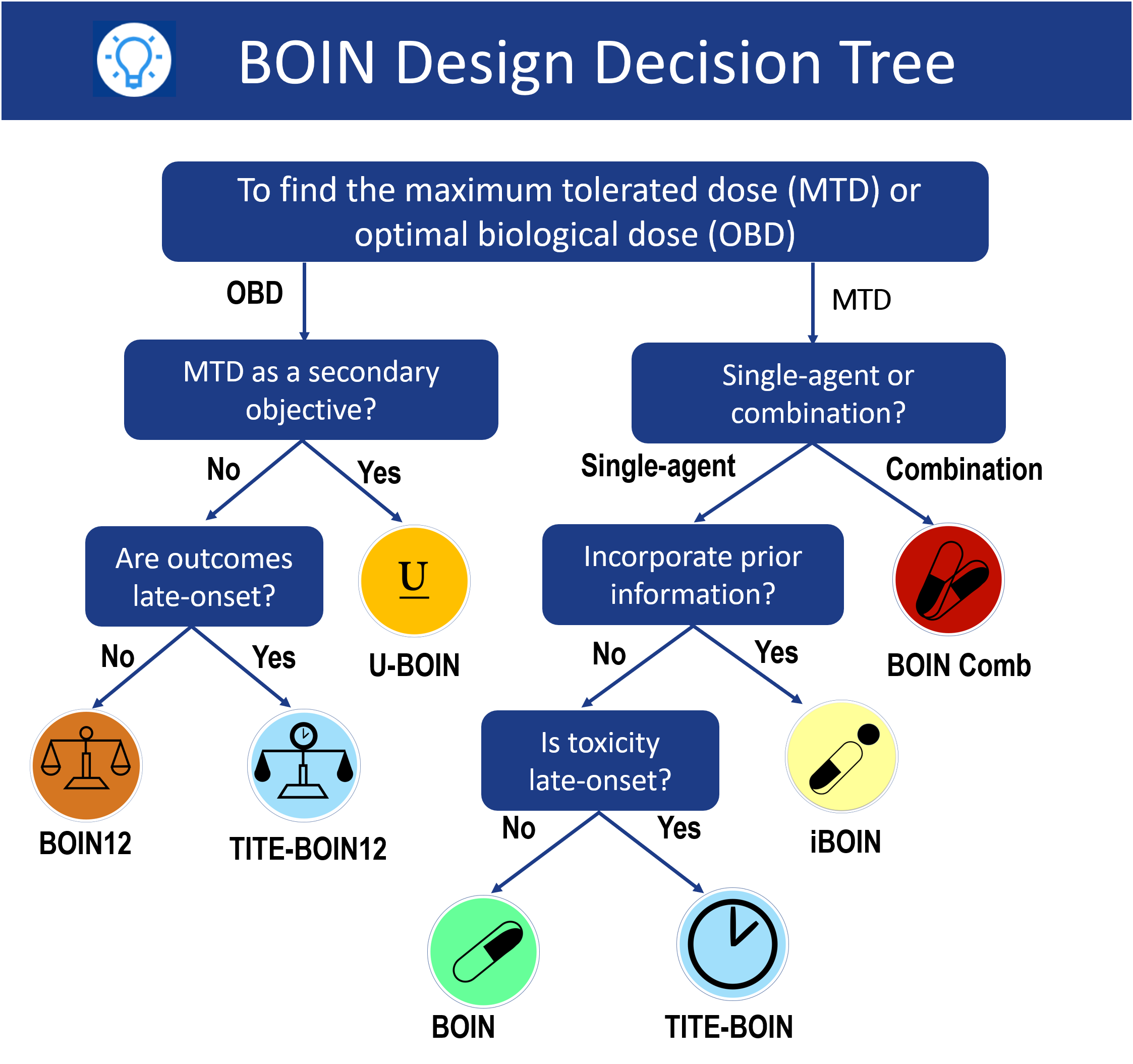
BOIN Suite: A Software Platform to Design and Implement Novel Early-Phase Clinical Trials | JCO Clinical Cancer Informatics

Metronomic Chemotherapy: Principles and Lessons Learned from Applications in the Treatment of Metastatic Prostate Cancer | SpringerLink

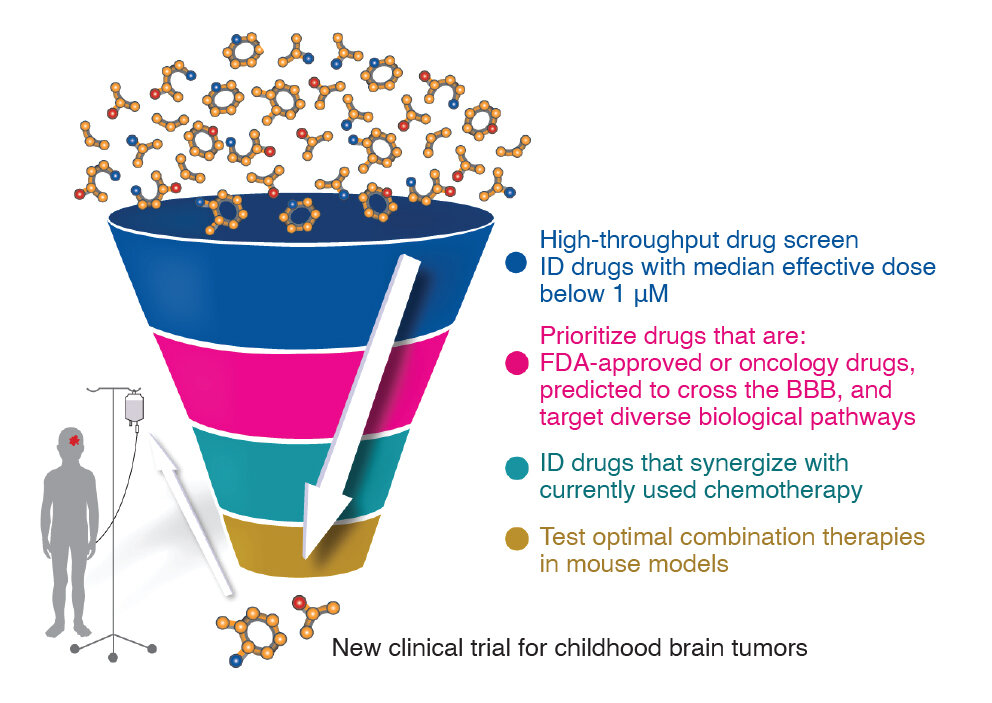
Prediction of drug candidates for clear cell renal cell carcinoma using a systems biology-based drug repositioning approach - eBioMedicine

Optimizing biologic therapy in IBD: how essential is therapeutic drug monitoring? | Nature Reviews Gastroenterology & Hepatology
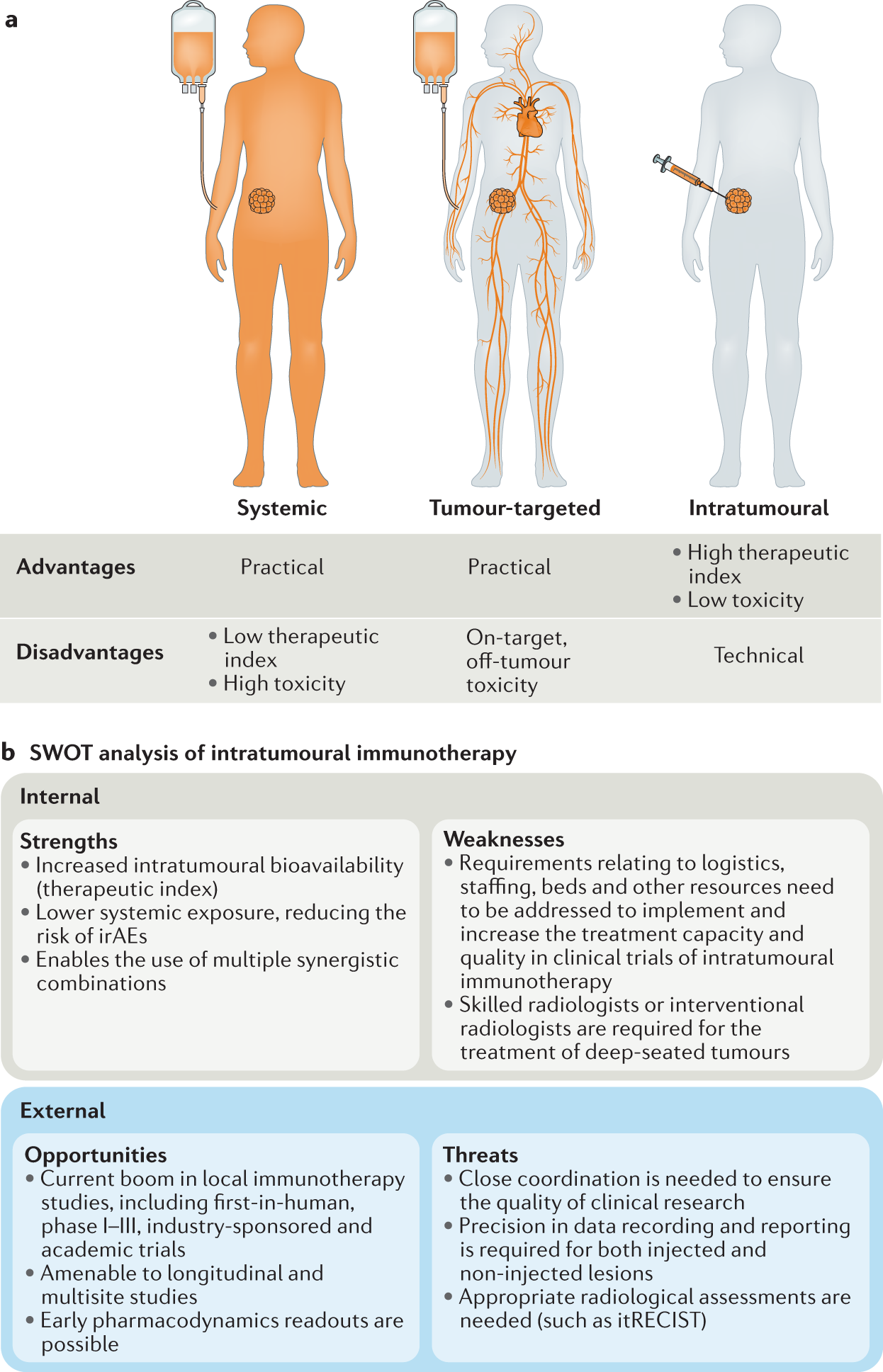
Intratumoural administration and tumour tissue targeting of cancer immunotherapies | Nature Reviews Clinical Oncology
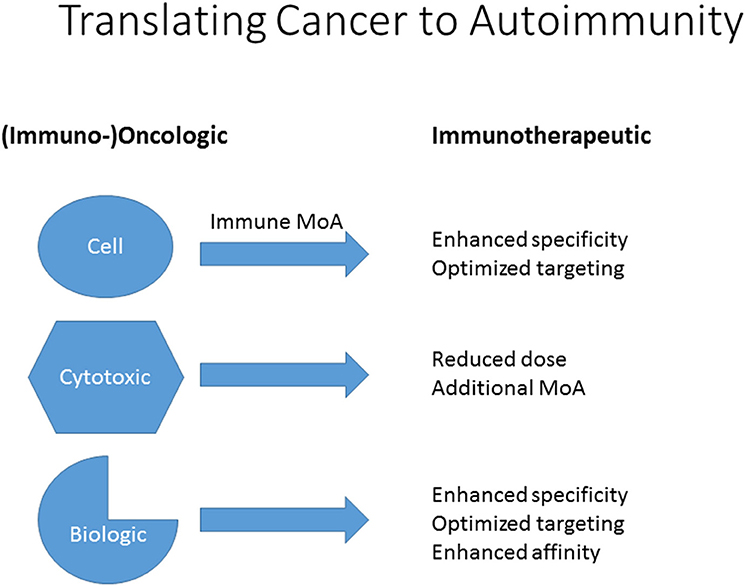
Frontiers | From Cancer to Immune-Mediated Diseases and Tolerance Induction: Lessons Learned From Immune Oncology and Classical Anti-cancer Treatment
Pharmacodynamic markers to support optimal biological dose selection in... | Download Scientific Diagram

Clinical efficacy of the optimal biological dose in early-phase trials of anti-cancer targeted therapies - ScienceDirect

Optimal biological dose: a systematic review in cancer phase I clinical trials | BMC Cancer | Full Text
Keyboard: A Bayesian Model-assisted Design R Package for Single- and Dual-agent Dose-Finding Early-phase Trials
PK/PD Mediated Dose Optimization of Emactuzumab, a CSF1R Inhibitor, in Patients With Advanced Solid Tumors and Diffuseâ•'Ty

Why do we conduct biological correlative studies for targeted agents? 1.We often have difficulty in determining the optimal dose/schedule in phase 1 to. - ppt download
Administration of Optimal Biological Dose and Schedule of Interferon Combined with Gemcitabine Induces Apoptosis in Tumor-associ

ELIZABETH GARRETT-MAYER (SOME SLIDES BY PAT LORUSSO OF KARMANOS CANCER INSTITUTE WAYNE STATE UNIVERSITY) Phase I Trials of Chemotherapy and Targeted Agents. - ppt download