Flat-dose nivolumab well-tolerated, active for advanced NSCLC in Asians | Latest news for Doctors, Nurses and Pharmacists | Oncology

ELCC 2022: Flat-dose nivolumab (NIVO) as second-line (2L) treatment (tx) in Asian patients (pts) with advanced non-small cell lung cancer (NSCLC): CheckMate 870 long-term results
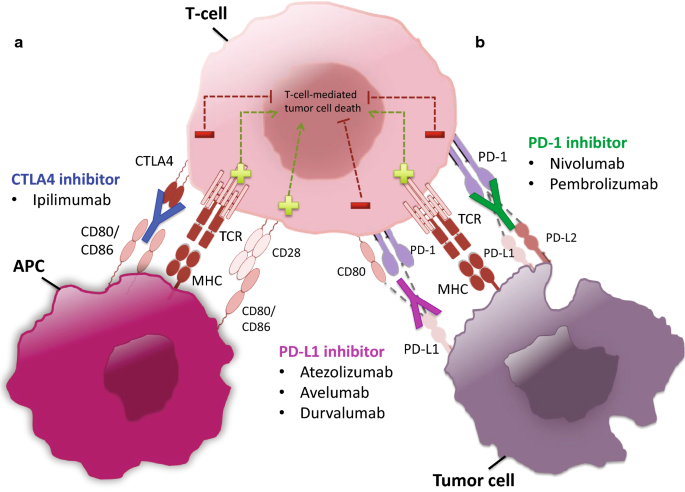
Nivolumab dose selection: challenges, opportunities, and lessons learned for cancer immunotherapy | Journal for ImmunoTherapy of Cancer
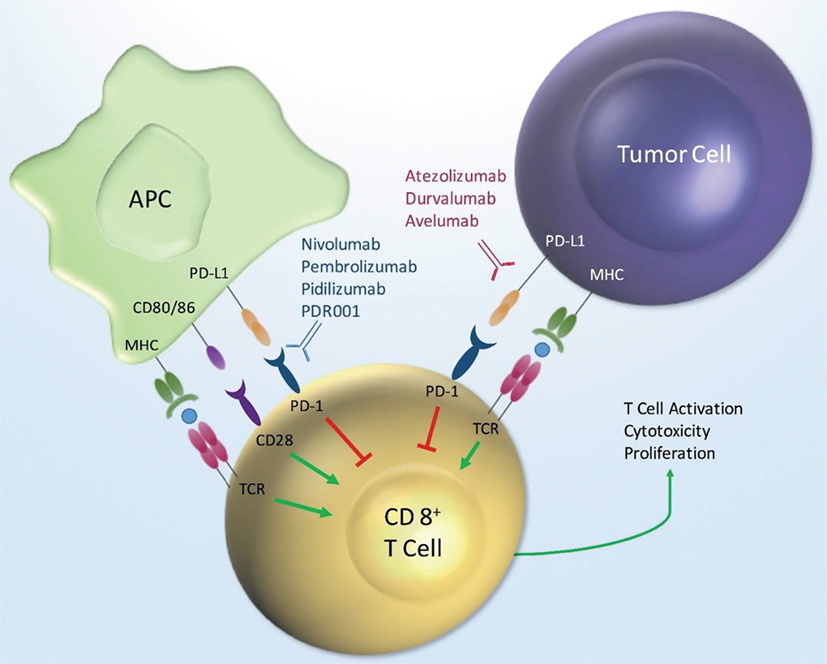
Frontiers | Incidence of Immune-Related Adverse Events with Program Death Receptor-1- and Program Death Receptor-1 Ligand-Directed Therapies in Genitourinary Cancers

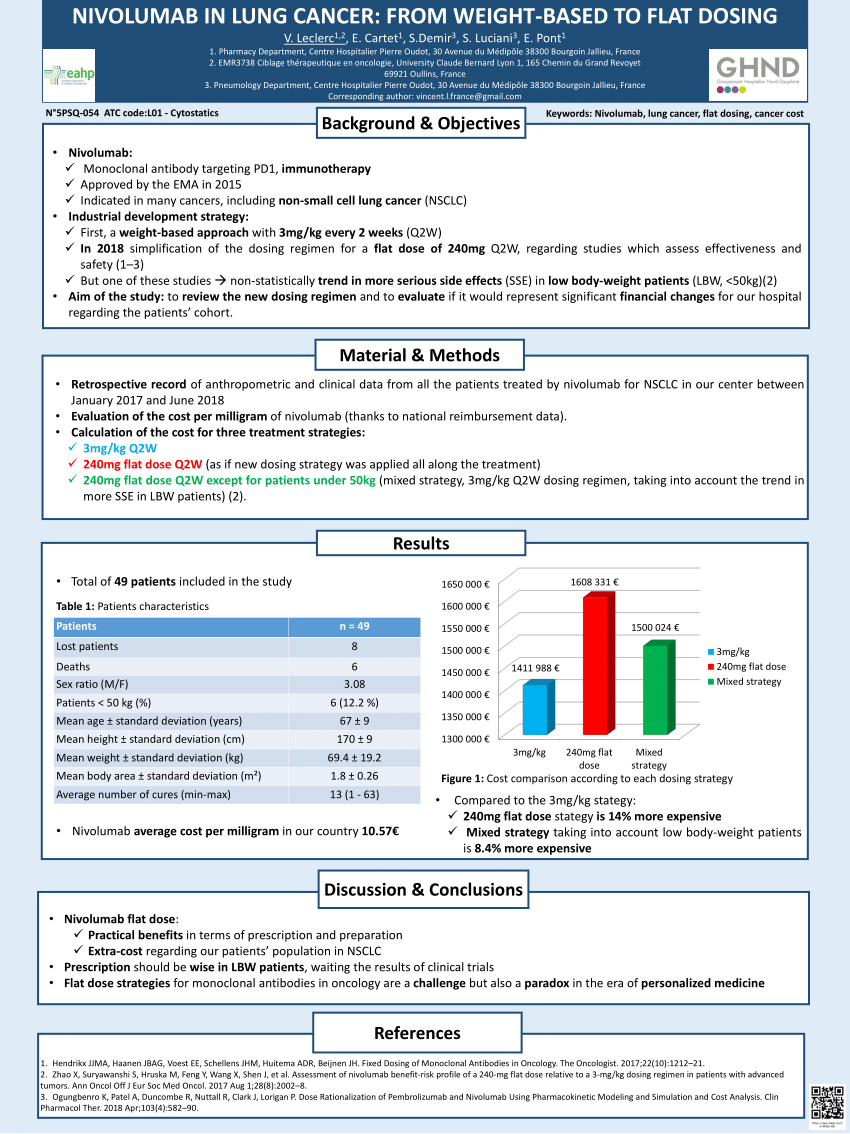
Alternative dosing strategies for immune checkpoint inhibitors to improve cost-effectiveness: a special focus on nivolumab and pembrolizumab - The Lancet Oncology
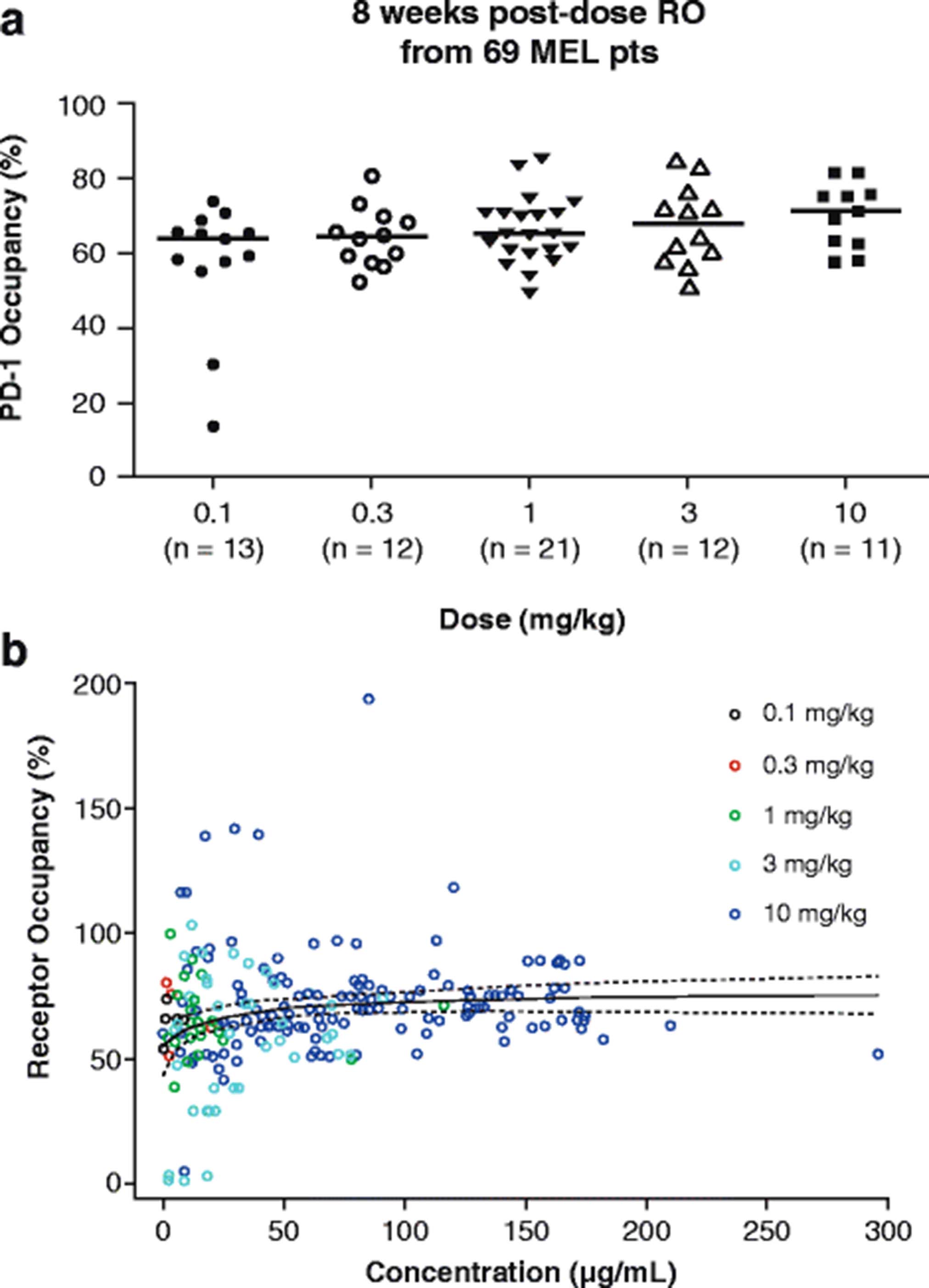
Nivolumab dose selection: challenges, opportunities, and lessons learned for cancer immunotherapy | Journal for ImmunoTherapy of Cancer
Assessment of nivolumab exposure and clinical safety of 480 mg every 4 weeks flat-dosing schedule in patients with cancer - Annals of Oncology

Efficacy of a Flat Low Dose of Nivolumab in Advanced Cancer: A Retrospective Case Series | Anticancer Research

Checkmate 649 1L Advanced or Metastatic Gastric, GEJ, & Esophogeal Carcinoma Dosing Schedule | OPDIVO® (nivolumab) + FOLFOX or CapeOX

Assessment of nivolumab exposure and clinical safety of 480 mg every 4 weeks flat-dosing schedule in patients with cancer - ScienceDirect

Analyses of Nivolumab Exposure and Clinical Safety Between 3-mg/kg Dosing and 240-mg Flat Dosing in Asian Patients with Advanced Renal Cell Carcinoma in the Real-World Clinical Setting - ScienceDirect

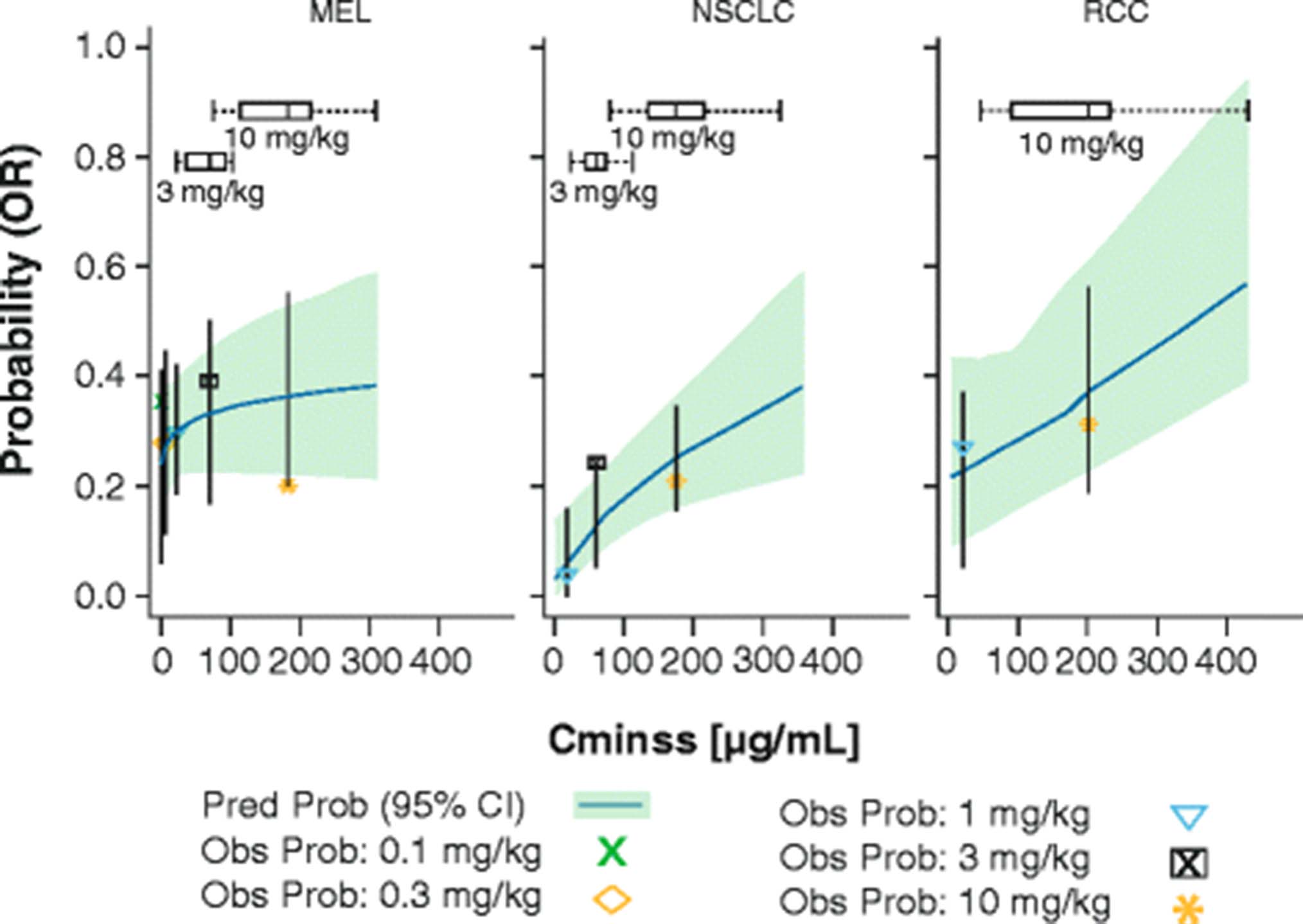
PDF) Assessment of nivolumab benefit-risk profile of a 240-mg flat dose relative to a 3-mg/kg dosing regimen in patients with advanced tumors

Benefit‐risk assessment of nivolumab 240 mg flat dose relative to 3 mg/kg Q2W regimen in Japanese patients with advanced cancers - Bei - 2020 - Cancer Science - Wiley Online Library

ELCC 2022: Flat-dose nivolumab (NIVO) as second-line (2L) treatment (tx) in Asian patients (pts) with advanced non-small cell lung cancer (NSCLC): CheckMate 870 long-term results