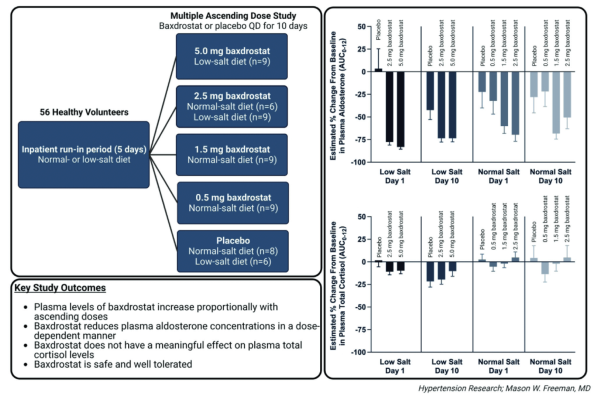
Safety, Tolerability, and Pharmacokinetics of Intravenous Oseltamivir: Single- and Multiple-Dose Phase I Studies with Healthy Volunteers | Antimicrobial Agents and Chemotherapy

Multiple-dose study. (A) Mean avatrombopag concentration time profiles... | Download Scientific Diagram
![PDF] Single and Multiple Ascending-dose Studies of Oral Delafloxacin: Effects of Food, Sex, and Age. | Semantic Scholar PDF] Single and Multiple Ascending-dose Studies of Oral Delafloxacin: Effects of Food, Sex, and Age. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/168f1250036d7761525361015074a1a817a3139b/7-Figure1-1.png)
PDF] Single and Multiple Ascending-dose Studies of Oral Delafloxacin: Effects of Food, Sex, and Age. | Semantic Scholar

Single- and Multiple-Dose Study To Determine the Safety, Tolerability, Pharmacokinetics, and Food Effect of Oral MRX-I versus Linezolid in Healthy Adult Subjects | Antimicrobial Agents and Chemotherapy

Study design. The first dose in the multiple-dose dose-escalation phase... | Download Scientific Diagram

Single and Multiple Ascending-dose Studies of Oral Delafloxacin: Effects of Food, Sex, and Age - ScienceDirect

Multiple Dose Pharmacokinetics and Safety of Sulcardine Sulfate in Healthy Chinese Male Subjects: An Open-Label Phase I Clinical Study | springermedizin.de

Chapter 22. Pharmacokinetics of Multiple Dosing | Handbook of Basic Pharmacokinetics… Including Clinical Applications, 7th Edition | PharmacyLibrary
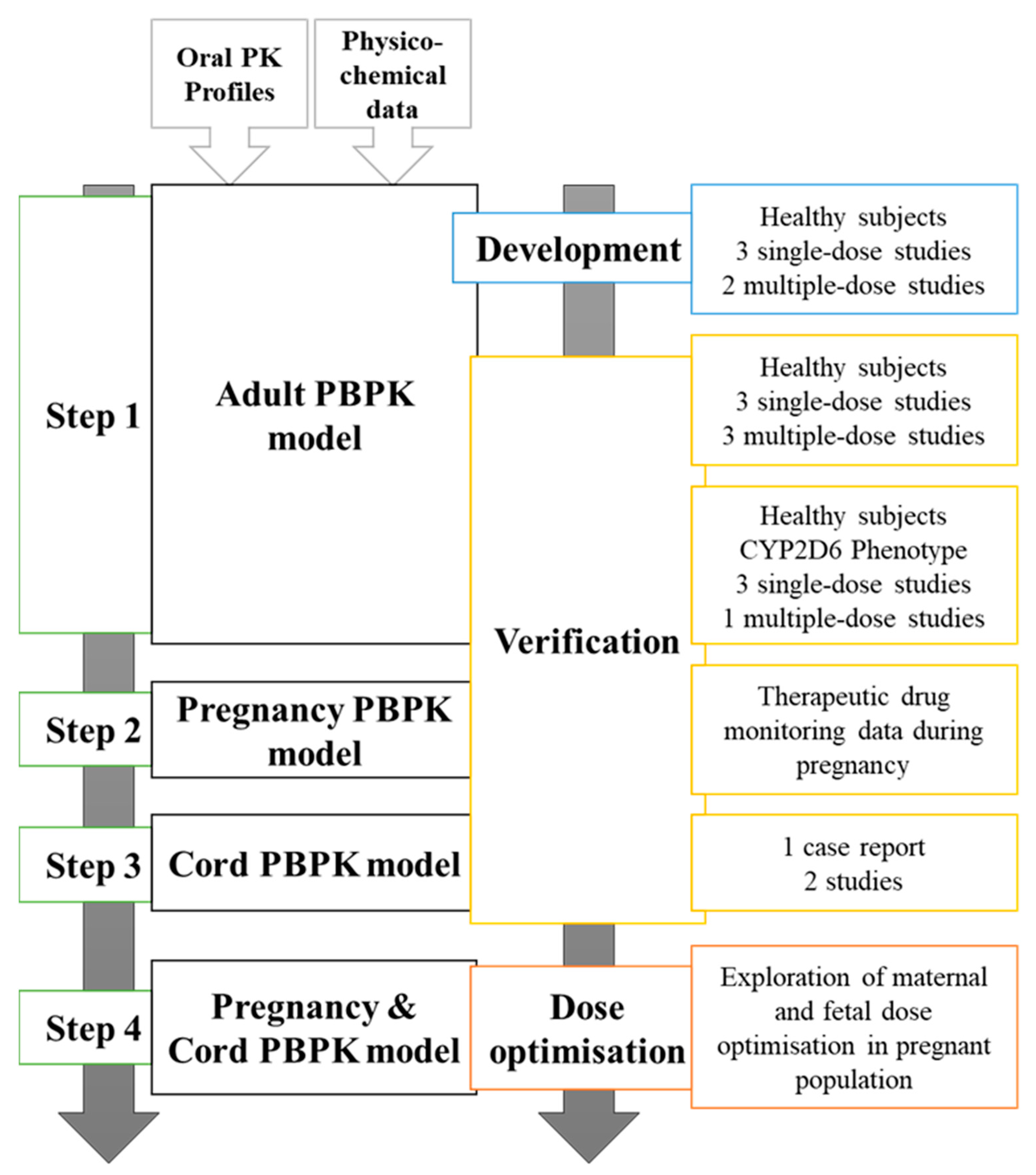
Metabolites | Free Full-Text | Optimising Fluvoxamine Maternal/Fetal Exposure during Gestation: A Pharmacokinetic Virtual Clinical Trials Study

A First‐in‐Human Phase 1 Randomized Single and Multiple Ascending Dose Study of RPh201 in Healthy Volunteers - Hazan - 2020 - Clinical Pharmacology in Drug Development - Wiley Online Library

Investigation on the need of multiple dose bioequivalence studies for prolonged-release generic products - ScienceDirect
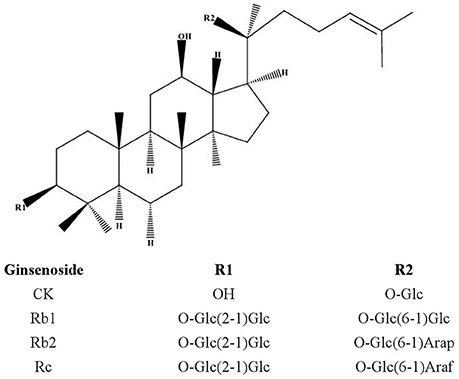
Frontiers | Single- and Multiple-Dose Trials to Determine the Pharmacokinetics, Safety, Tolerability, and Sex Effect of Oral Ginsenoside Compound K in Healthy Chinese Volunteers

Chapter 22. Pharmacokinetics of Multiple Dosing | Handbook of Basic Pharmacokinetics… Including Clinical Applications, 7th Edition | PharmacyLibrary
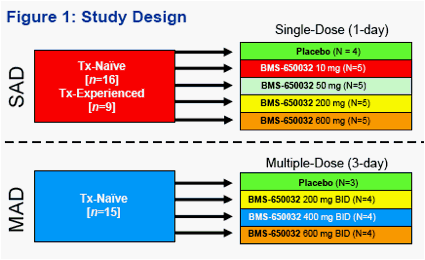
Safety, Tolerability, Pharmacokinetics and Antiviral Activity following Single- and Multiple-Dose Administration of BMS-650032, a Novel HCV NS3 Inhibitor, in Subjects with Chronic Genotype 1 HCV Infection
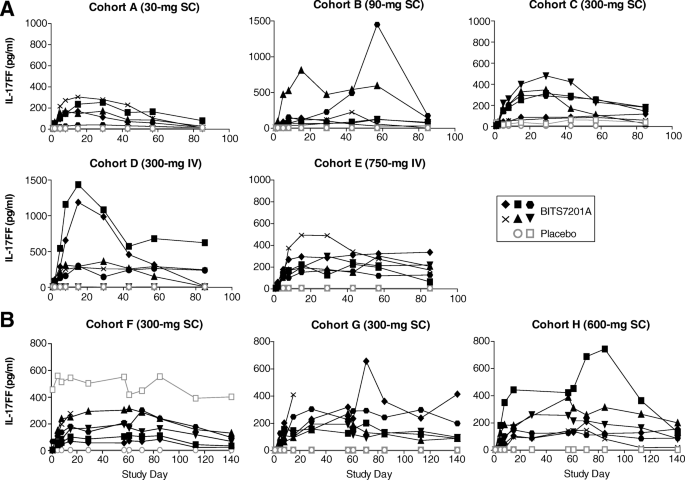
A phase I, randomized, observer-blinded, single and multiple ascending-dose study to investigate the safety, pharmacokinetics, and immunogenicity of BITS7201A, a bispecific antibody targeting IL-13 and IL-17, in healthy volunteers | BMC Pulmonary

Pharmacokinetics of RP5063 Following Single Doses to Normal Healthy Volunteers and Multiple Doses Over 10 Days to Stable Schizophrenic Patients - Cantillon - 2018 - Clinical and Translational Science - Wiley Online Library

Multiple-Ascending-Dose Phase 1 Clinical Study of the Safety, Tolerability, and Pharmacokinetics of CRS3123, a Narrow-Spectrum Agent with Minimal Disruption of Normal Gut Microbiota | Antimicrobial Agents and Chemotherapy