
Impact of Changing Regulations and the Dynamic Nature of European Risk Management Plans for Human Medicines on the Lifecycle of Safety Concerns | SpringerLink
Der Risikomanagementplan: Neufokussierung durch die zweite Revision des Moduls V des EU-Leitfadens zur Guten Pharmakovigilanz

The Pharmacovigilance Medical Writer: Medical Writer, Project Manager, Regulatory Expert - Trilogy Writing & Consulting GmbH

DOC) Response to Consultation from European Medicines Agency: Good Pharmacovigilance Practice Module V – Risk management systems' (EMA/838713/2011) | Theo Raynor - Academia.edu
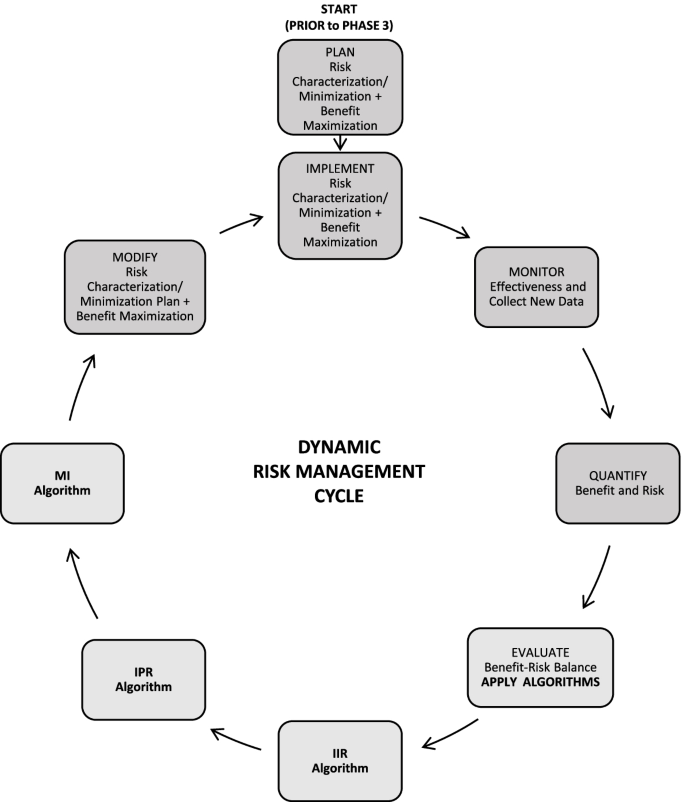






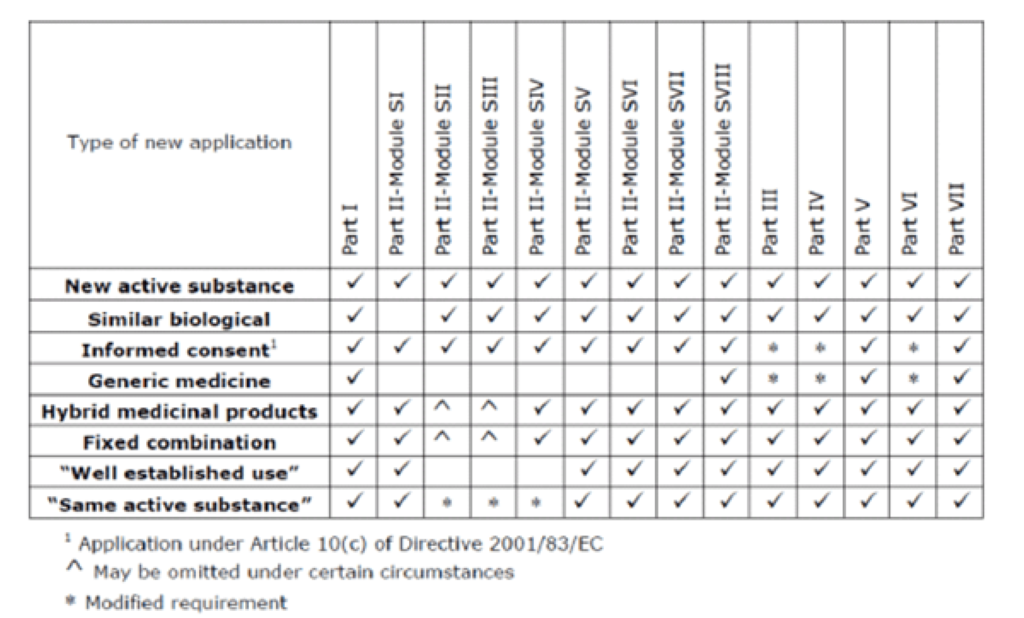



![EMA EXPECTATION WITH THE REVISED GUIDELINE OF RISK MANAGEMENT PLAN [GVP MODULE V] | NUJPS EMA EXPECTATION WITH THE REVISED GUIDELINE OF RISK MANAGEMENT PLAN [GVP MODULE V] | NUJPS](http://www.nujps.com/wp-content/uploads/2022/02/32.jpg)



