Frontiers | Evaluation of Information in Summaries of Product Characteristics (SmPCs) on the Use of a Medicine in Patients With Hepatic Impairment

An Analysis of the Fixed-Dose Combinations Authorized by the European Union, 2009-2014: A Focus on Benefit-Risk and Clinical Development Conditions - Dominik Sawicki-Wrzask, Mikael Thomsen, Ole J. Bjerrum, 2015

STRATEGIC APPROACHES OF UTILIZING BA/BE STUDIES TO OBTAIN APPROVAL FROM DIFFERENT REGULATORY AGENCIES FOR FIXED DOSE COMBINATION - BioPharma Services

EMA accepts Otsuka and Astex's marketing authorisation application for acute myeloid leukaemia treatment - PMLiVE
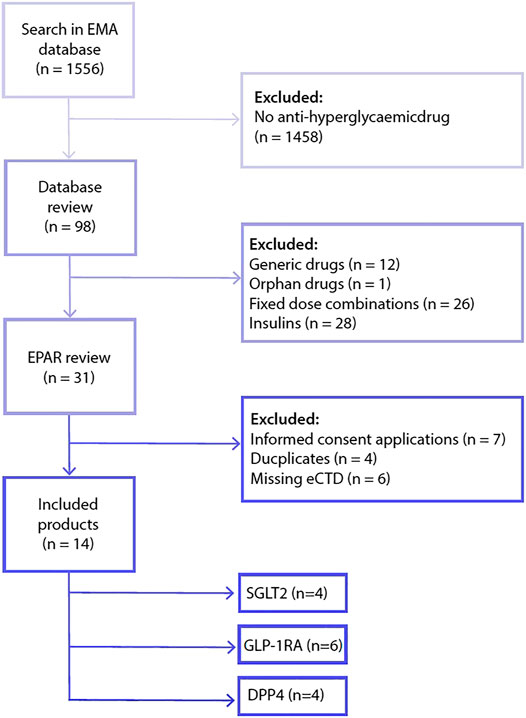
Frontiers | A Review of the Dose Justification of Phase 3 Trials to Regulatory Authorities for Drugs Intended for the Treatment of Type 2 Diabetes in Europe

Oral Fixed-Dose Combination Pharmaceutical Products: Industrial Manufacturing Versus Personalized 3D Printing | SpringerLink

An Analysis of the Fixed-Dose Combinations Authorized by the European Union, 2009-2014: A Focus on Benefit-Risk and Clinical Development Conditions - Dominik Sawicki-Wrzask, Mikael Thomsen, Ole J. Bjerrum, 2015

Guidance for pharmaceutical companies on how to prepare and review summaries of product characteristics (SmPCs) for human medicines. - ESPL Regulatory Consulting

Pharmaceutical application and development of fixed-dose combination: dosage form review | SpringerLink

PDF) A Comparative Review of Waivers Granted in Pediatric Drug Development by FDA and EMA from 2007-2013

Topical fixed-dose combinations: Current in vitro methodologies for pre-clinical development - ScienceDirect

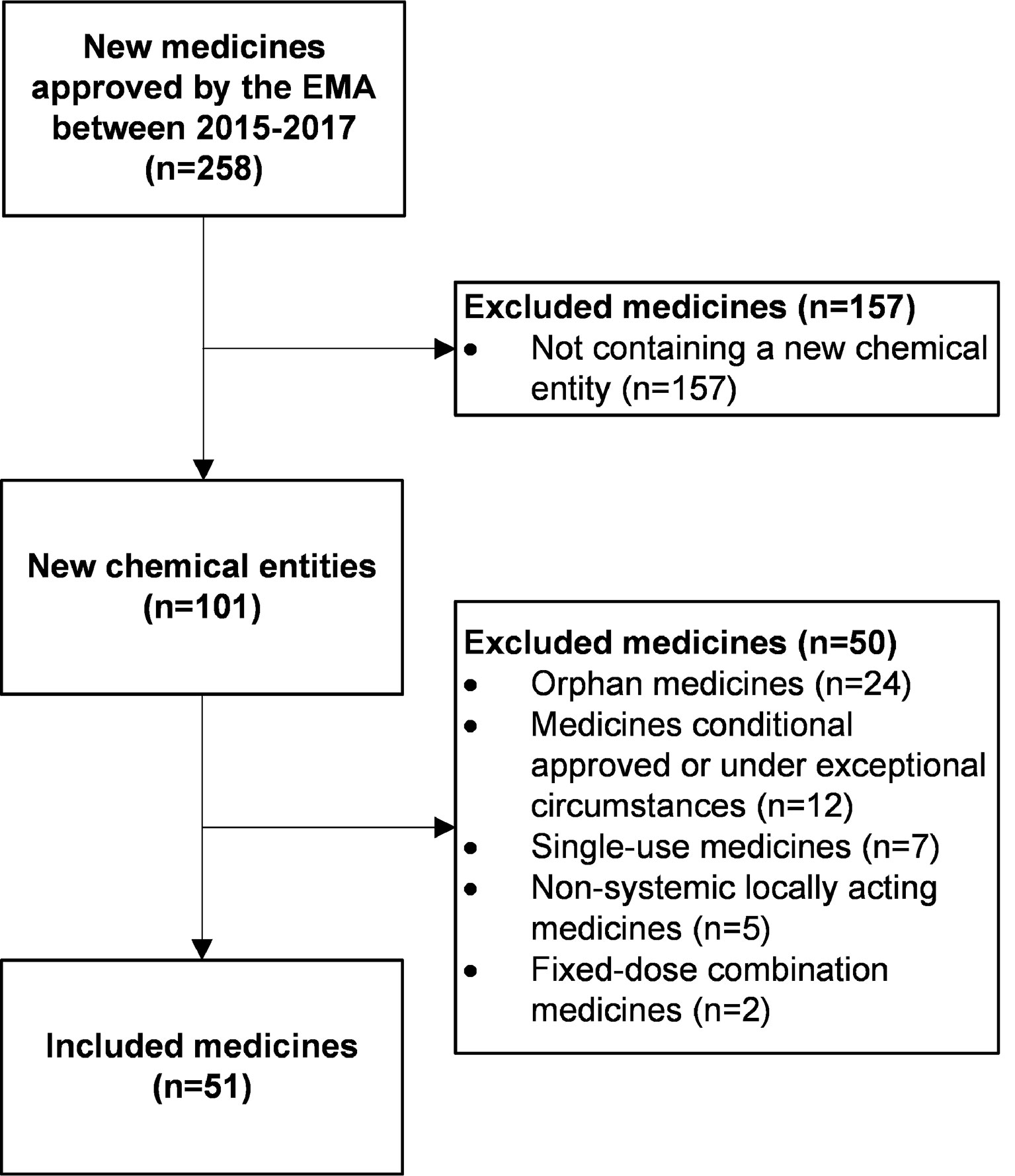
The European Medicines Agency's approval of new medicines for type 2 diabetes - Blind - 2018 - Diabetes, Obesity and Metabolism - Wiley Online Library
Use of Fixed Dose Combination (FDC) Drugs in India: Central Regulatory Approval and Sales of FDCs Containing Non-Steroidal Anti-Inflammatory Drugs (NSAIDs), Metformin, or Psychotropic Drugs | PLOS Medicine

EU Medicines Agency on Twitter: "What is EMA's advice on the use of non-steroidal anti-inflammatories like #ibuprofen for #COVID19? #NSAIDs #SafetyOfMedicines 👉https://t.co/qFidQE4e3w https://t.co/S7YVHRFhlF" / Twitter


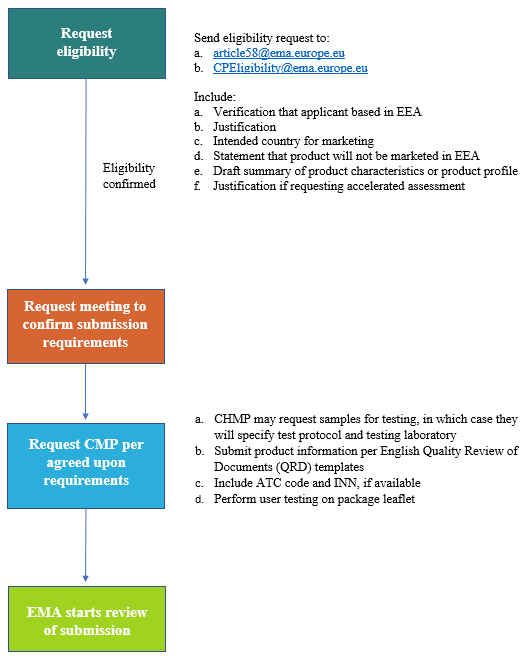

![PDF] Fixed dose combination product : A Review | Semantic Scholar PDF] Fixed dose combination product : A Review | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f43d4f1c5e14ec71949aed625321a7b65a618eb0/8-Table3-1.png)