AstraZeneca pledges more Evusheld doses to US, bringing its antibody supply deal to $855M | Fierce Pharma

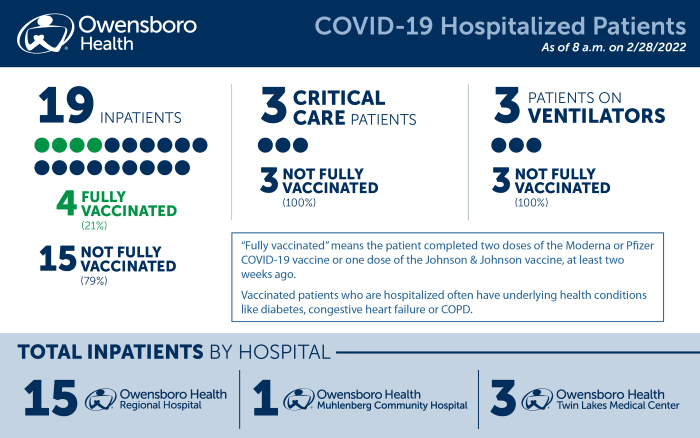
RCT finds single dose of Evusheld reduces progression of COVID-19 and mortality - Hospital Pharmacy EuropeHospital Pharmacy Europe
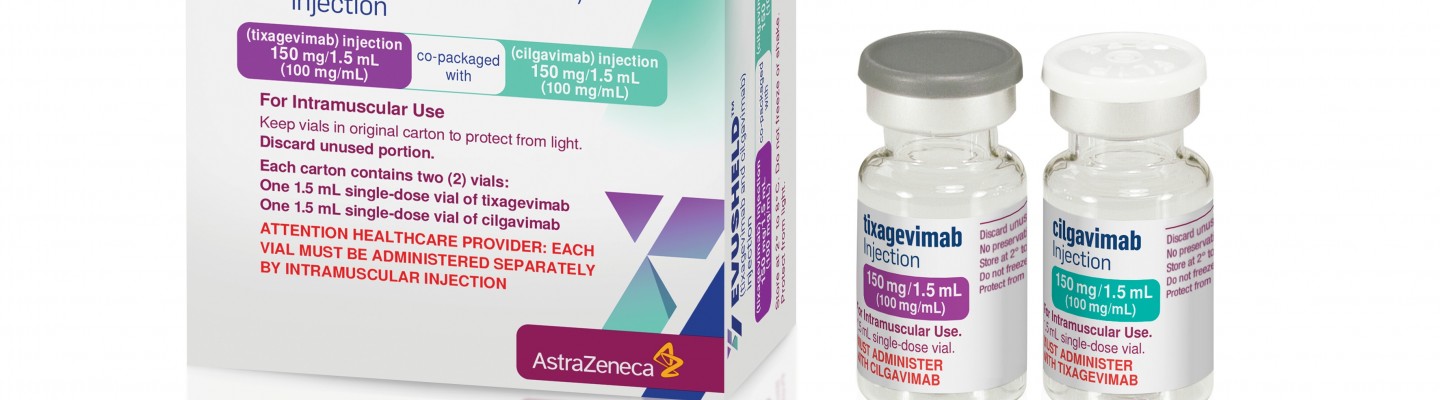
EVUSHELD (formerly AZD7442) long-acting antibody combination authorized for emergency use in the US for pre-exposure prophylaxis (prevention) of COVID-19 | Business Wire
Important notice for MS patients regarding Evusheld | Multiple Sclerosis Society of NZMultiple Sclerosis Society of NZ