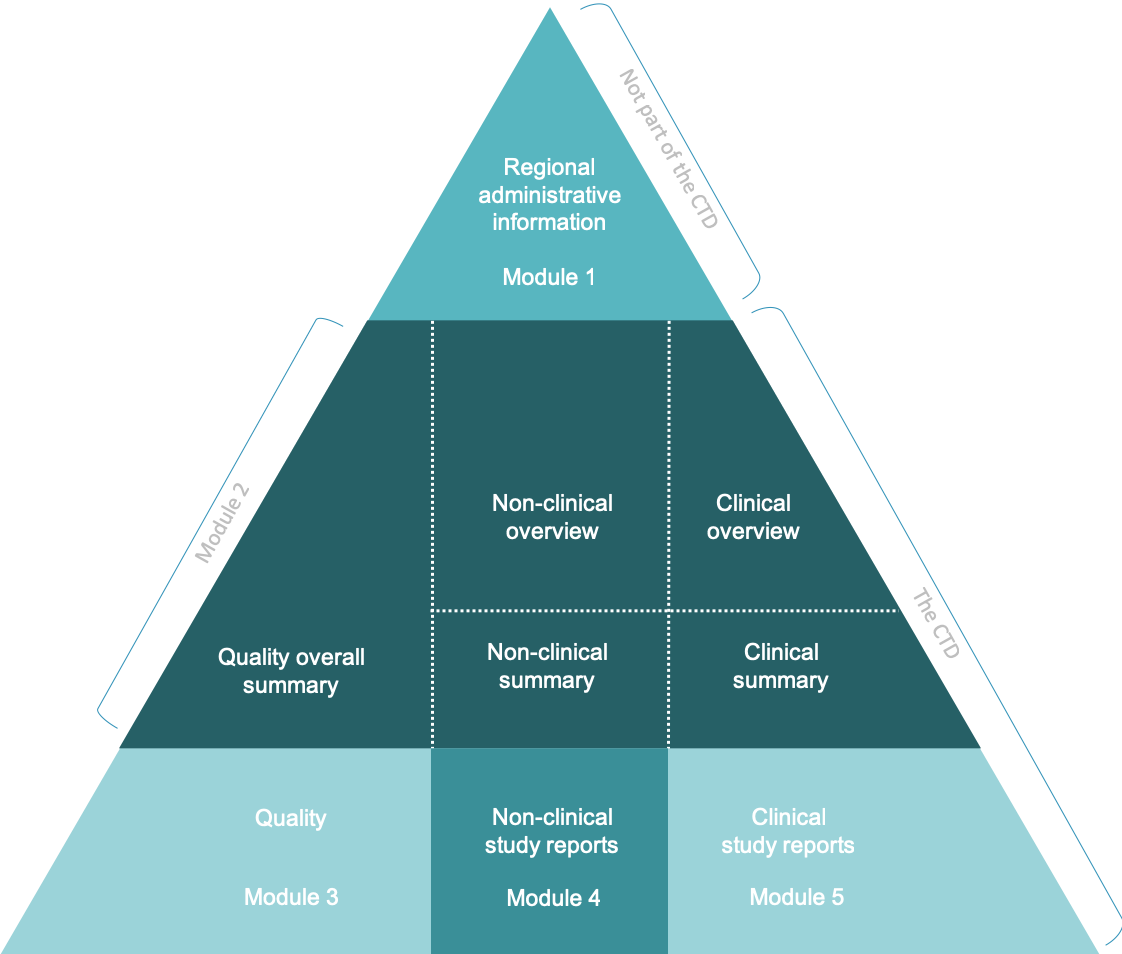
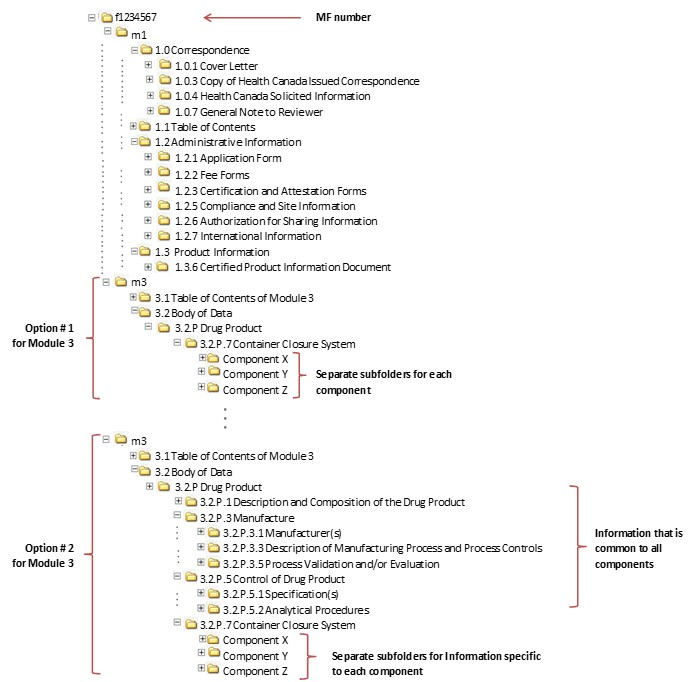
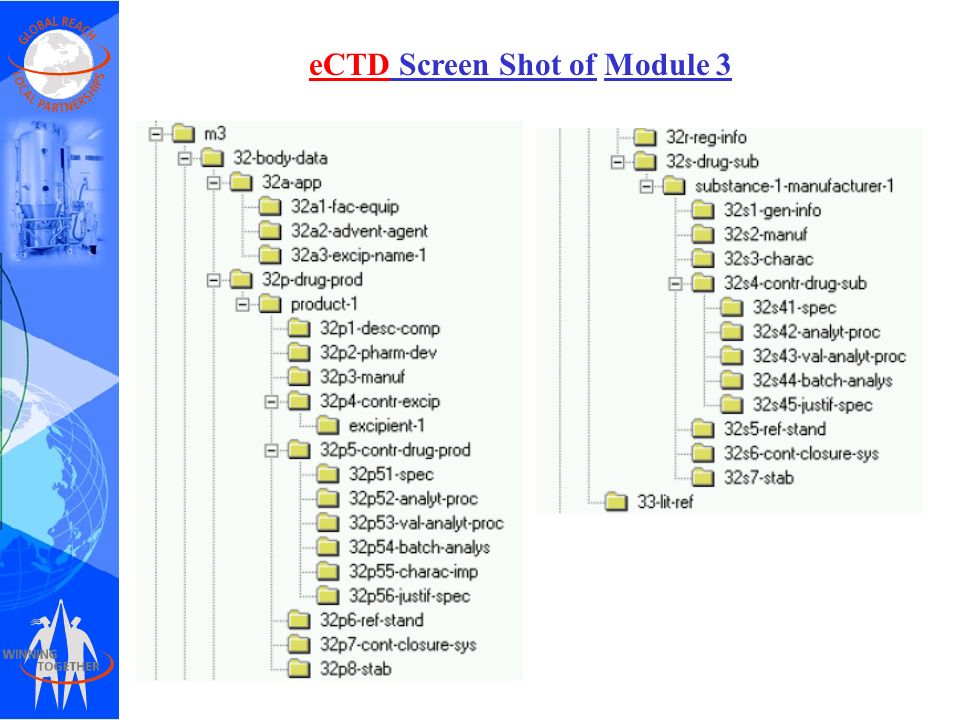
Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substan
![PDF] Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substances in Europe | Semantic Scholar PDF] Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substances in Europe | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/30903f1fc51c4917a2877b9cf3756ccc7fc6425a/16-Table2-1.png)
PDF] Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substances in Europe | Semantic Scholar
![PDF] Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substances in Europe | Semantic Scholar PDF] Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substances in Europe | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/30903f1fc51c4917a2877b9cf3756ccc7fc6425a/11-Table1-1.png)