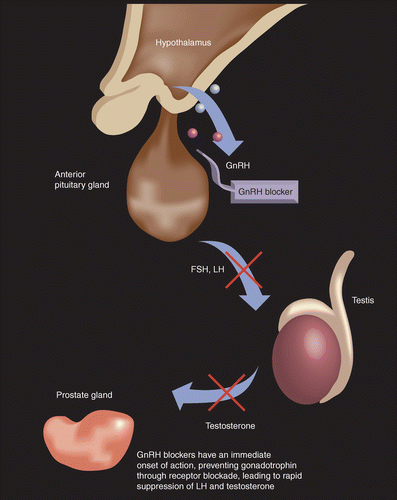
Efficacy and safety of 3‐month dosing regimen of degarelix in Japanese subjects with prostate cancer: A phase III study - Ozono - 2018 - Cancer Science - Wiley Online Library

These highlights do not include all the information needed to use FIRMAGON safely and effectively. See full prescribing information for FIRMAGON. FIRMAGON® (degarelix for injection) for subcutaneous use Initial U.S. Approval: 2008
.jpg)
COVID-19 Drugs: Phase Two Clinical Trial Of Degarelix Launched To Treat Only Male COVID-19 Patients - Thailand Medical News
![PDF] Efficacy and safety of a 3-month dosing regimen of degarelix in Japanese patients with prostate cancer: a phase II maintenance-dose-finding study | Semantic Scholar PDF] Efficacy and safety of a 3-month dosing regimen of degarelix in Japanese patients with prostate cancer: a phase II maintenance-dose-finding study | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f0f9acba807f066d8d2e5a43439569d8a02f1697/2-Figure1-1.png)
PDF] Efficacy and safety of a 3-month dosing regimen of degarelix in Japanese patients with prostate cancer: a phase II maintenance-dose-finding study | Semantic Scholar

Effect of Degarelix, a Gonadotropin-Releasing Hormone Receptor Antagonist for the Treatment of Prostate Cancer, on Cardiac Repolarisation in a Randomised, Placebo and Active Comparator Controlled Thorough QT/QTc Trial in Healthy Men

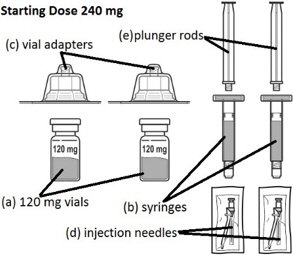
Firmagon® Starting Dose Antineoplastic Agent Degarelix Acetate 120 mg Subcutaneous Injection Kit 2 X 120 mg - Suprememed

The efficacy and safety of degarelix: a 12‐month, comparative, randomized, open‐label, parallel‐group phase III study in patients with prostate cancer - Klotz - 2008 - BJU International - Wiley Online Library