
Dr Jitendra Singh advocates integrated training modules in governance for Central Training Institutions
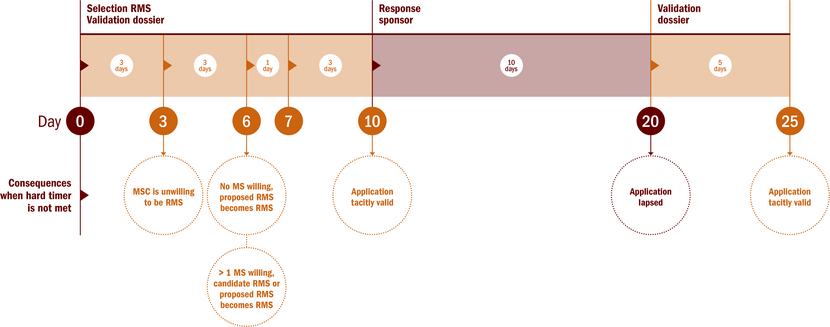
Validation phase: general information | Investigators | The Central Committee on Research Involving Human Subjects

📢 We've developed this new course to help you get a clear overview of the Clinical Trial Regulation 536/2014 (CTR) and help you to implement it in your... | By European Centre









