The Challenge of CTD Submissions and Responding to Questions from the Authorities - Trilogy Writing & Consulting GmbH

An Introduction to Integrated Summary of Safety and Integrated Summary of Effectiveness (ISS and ISE) - Quantics Biostatistics

ICH M4Q Common technical document for the registration of pharmaceuticals for human use – Introduction – Orioled Hub
Exposé zum Thema: eCTD - Neue Wege der elektronischen Arzneimittelzulassung und die Vernetzung elektronischer regulatorischer P

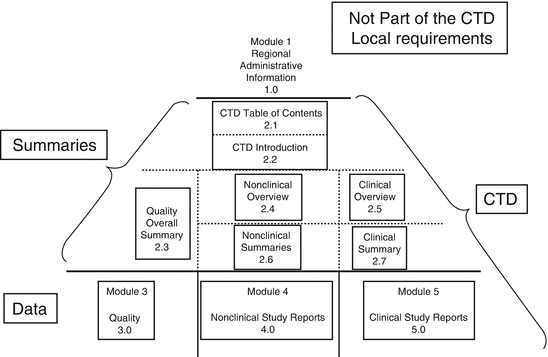
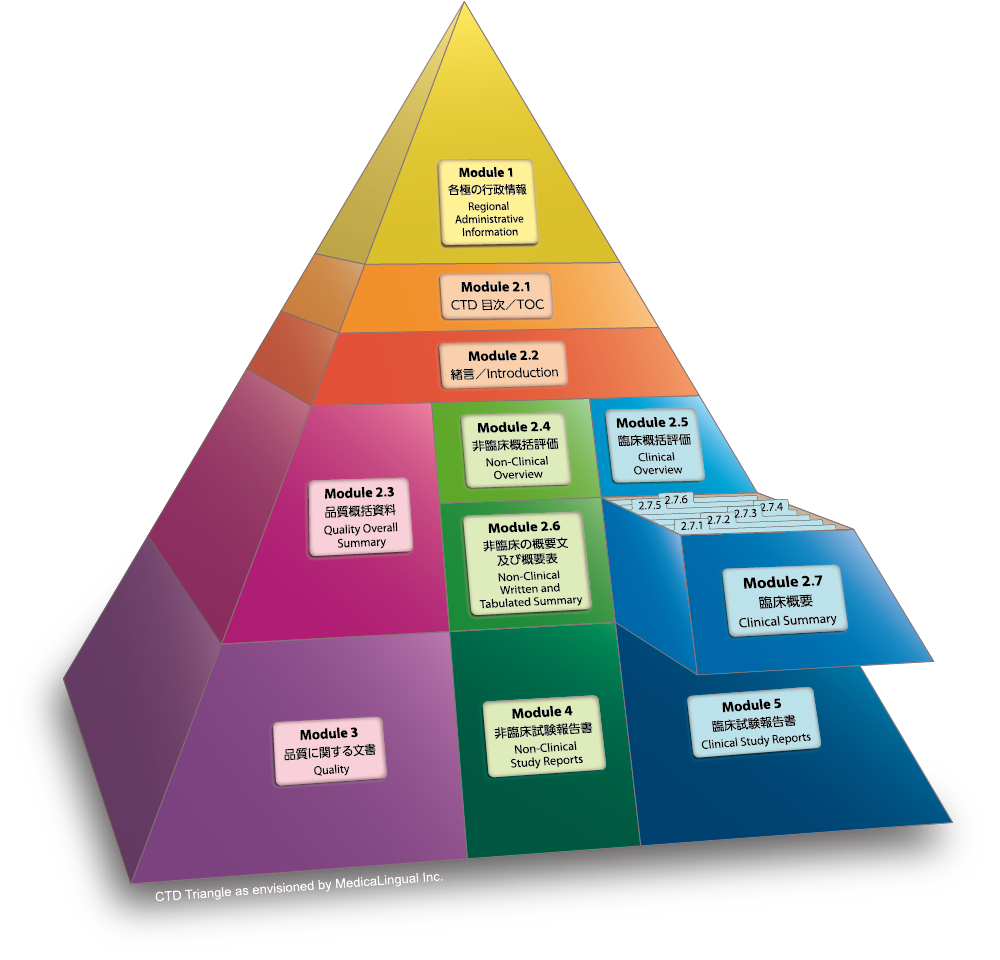
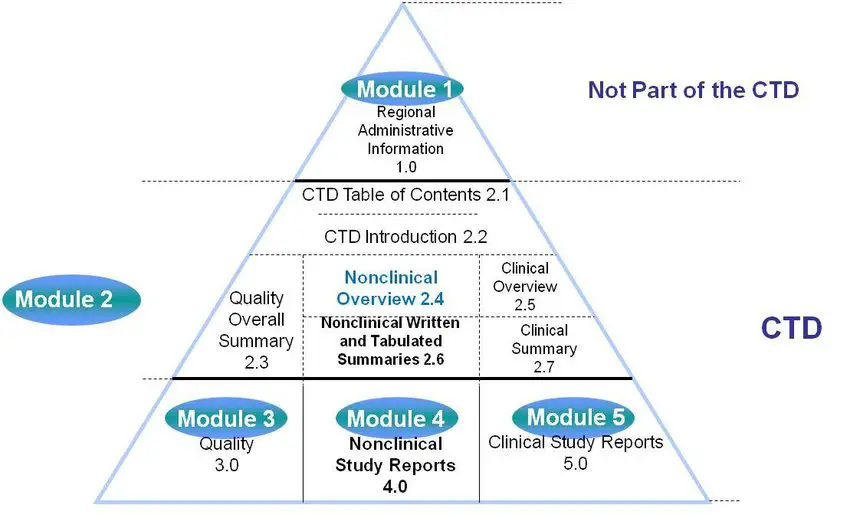
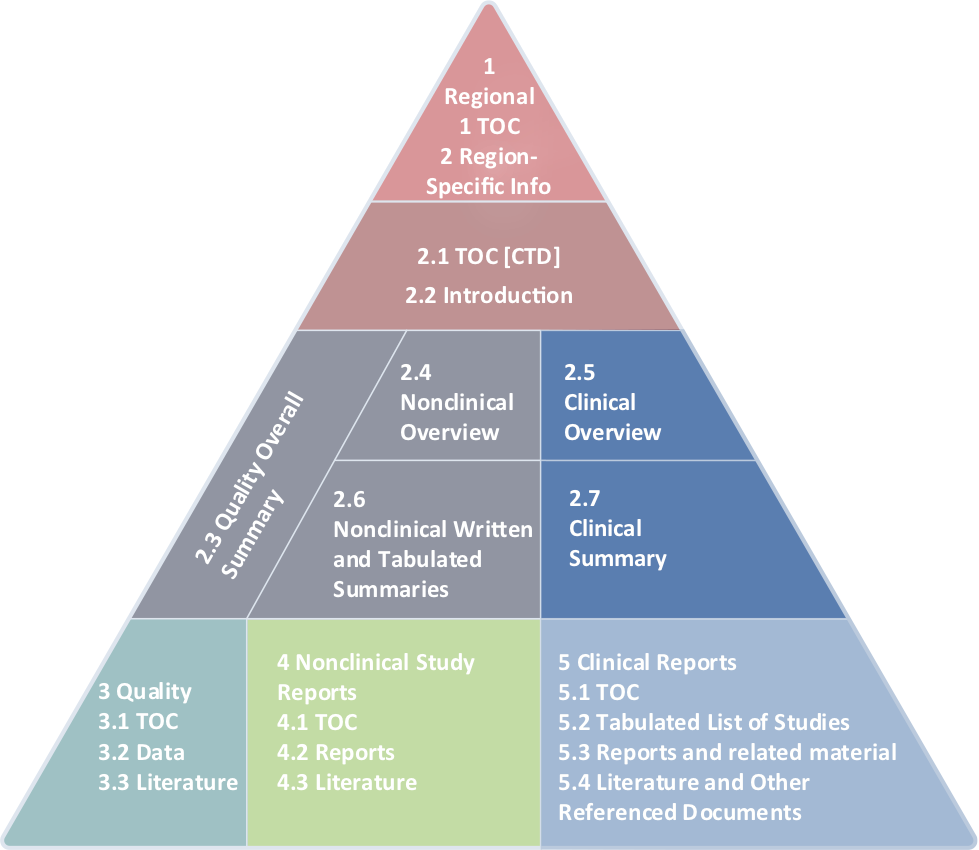
Schematic representation of the five modules in the Common Technical... | Download Scientific Diagram
WHO Guidelines on submission of documentation for the pilot procedure for prequalification of similar biotherapeutic products fo
Potential to Simplify the Writing of Submission Documents: Evaluation of Publicly Available Module 2 Documents in Drug Submissio