Potential to Simplify the Writing of Submission Documents: Evaluation of Publicly Available Module 2 Documents in Drug Submissio

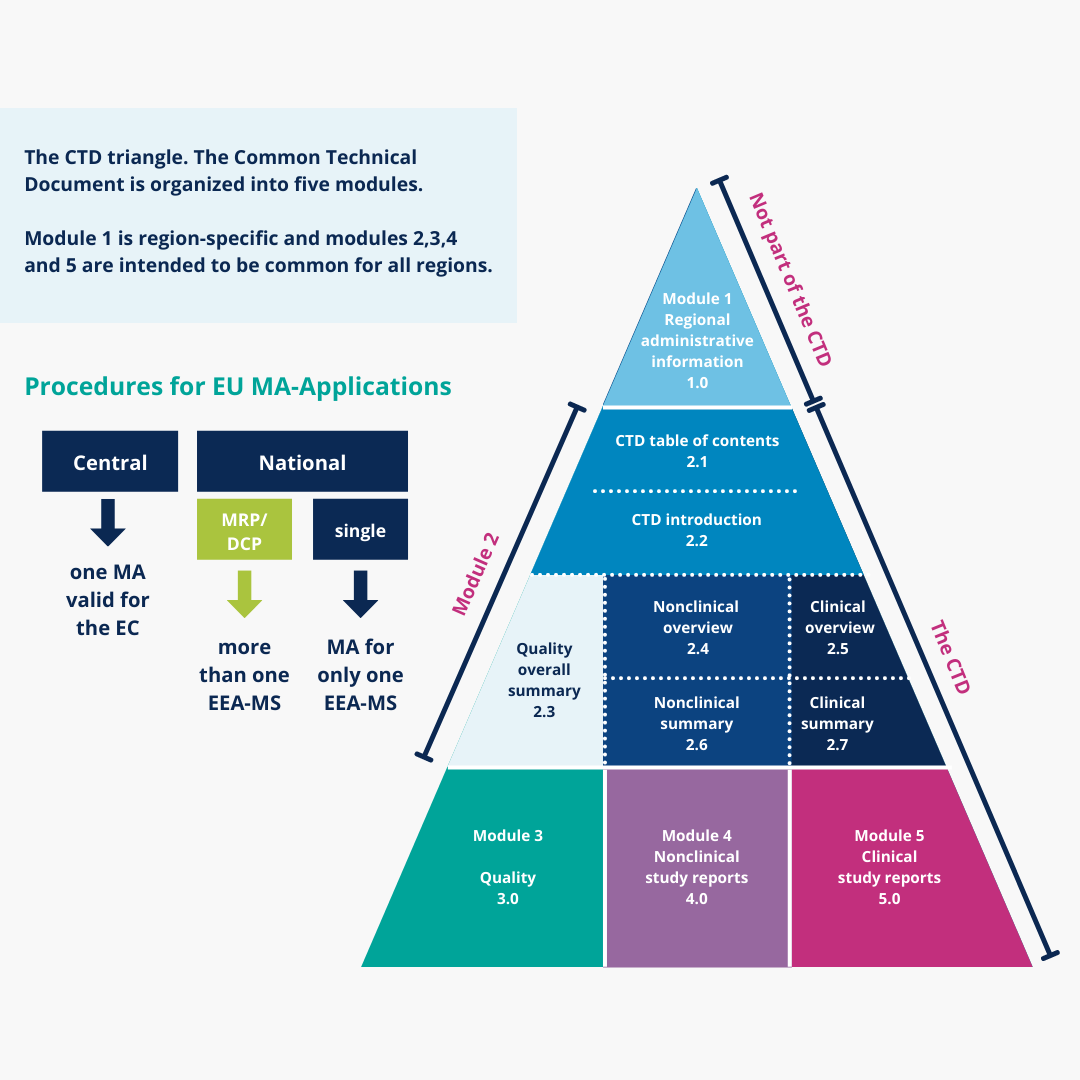
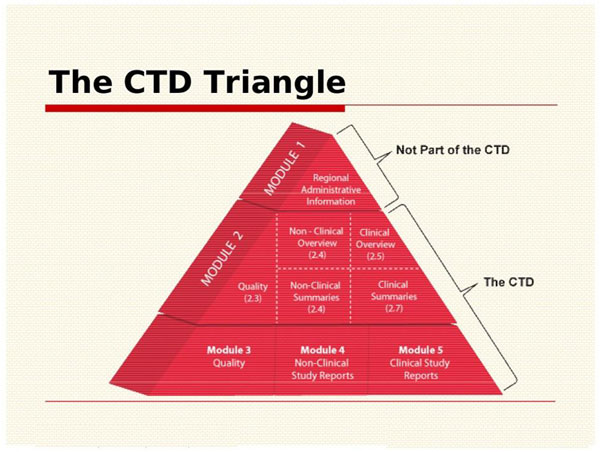
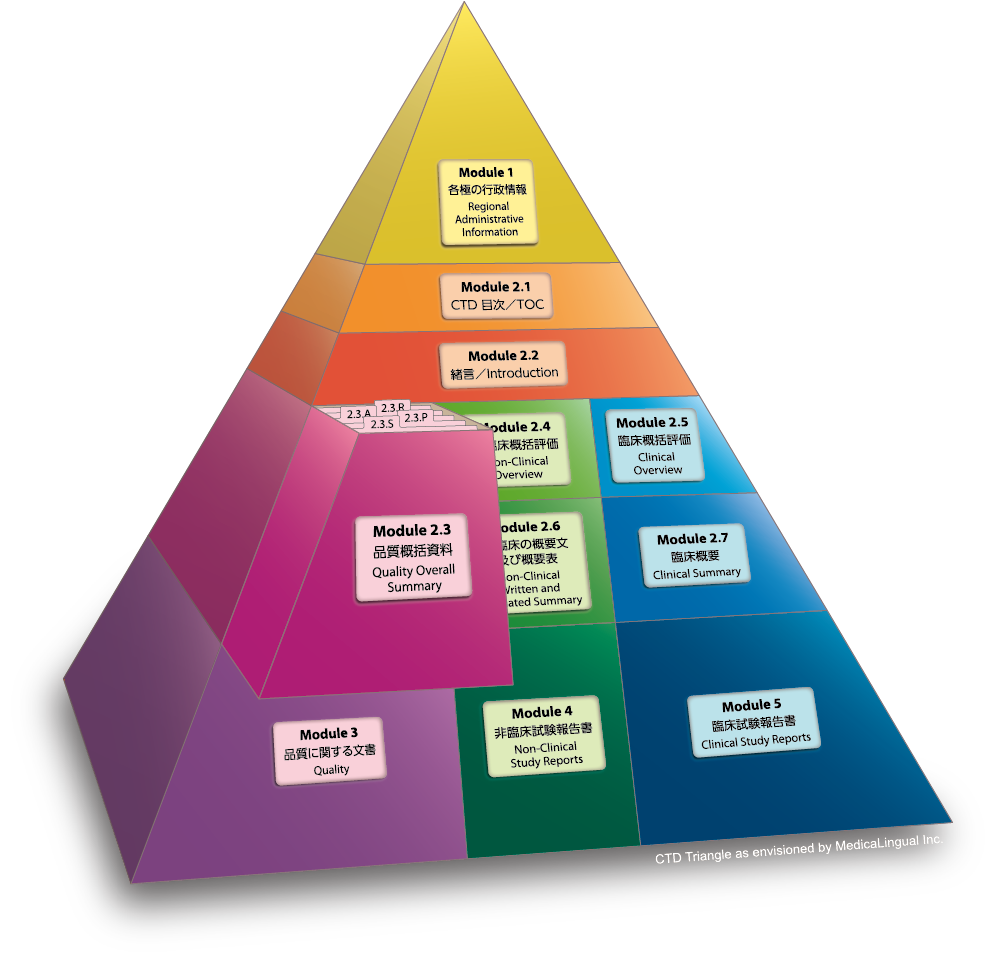
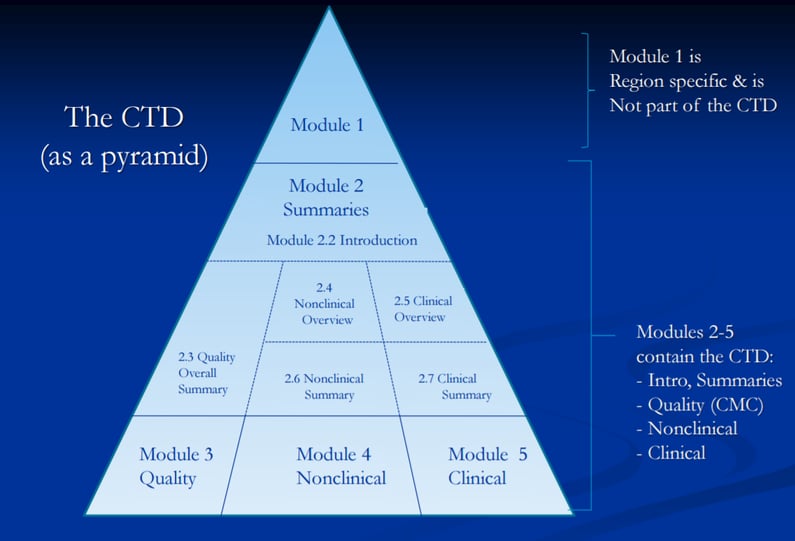
1 COMMON TECHNICAL DOCUMENT / ORIGIN OF CTD… ICH EWG CTD WAS OFFICIALLY SIGNED OFF IN NOVEMBER 2000, AT 5 TH ICH CONFERENCE; SAN DIEGO,CALIFORNIA. - ppt download
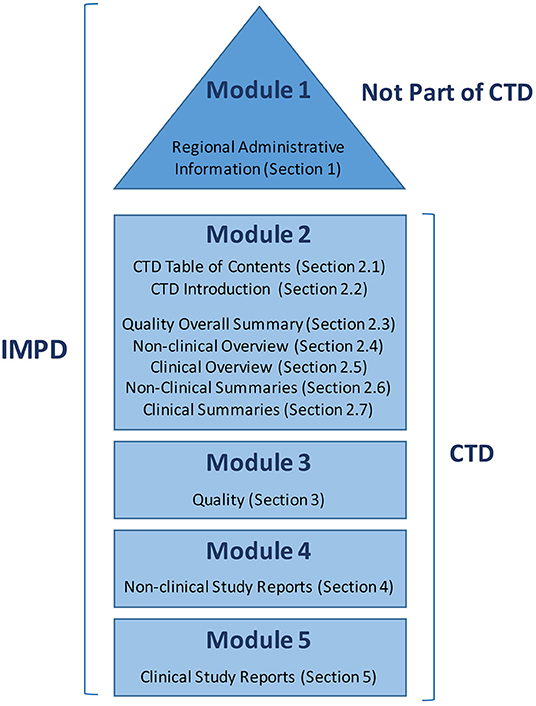
Frontiers | Transitioning From Preclinical Evidence to Advanced Therapy Medicinal Product: A Spanish Experience

Diagrammatic representation of the organization of follow-on biologics... | Download Scientific Diagram
Guideline on the use of the CTD format in the preparation of a registration application for traditional herbal medicinal product
Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substan
WHO Guidelines on submission of documentation for the pilot procedure for prequalification of similar biotherapeutic products fo

PDF) PREPARATION AND REVIEW OF CHEMISTRY, MANUFACTURING AND CONTROL (CMC) SECTIONS OF CTD DOSSIER FOR MARKETING AUTHORIZATION
Between Standardisation and Flexibility – Defining Granularity of the eCTD Module 3.2.S for Different Types of Drug Substan