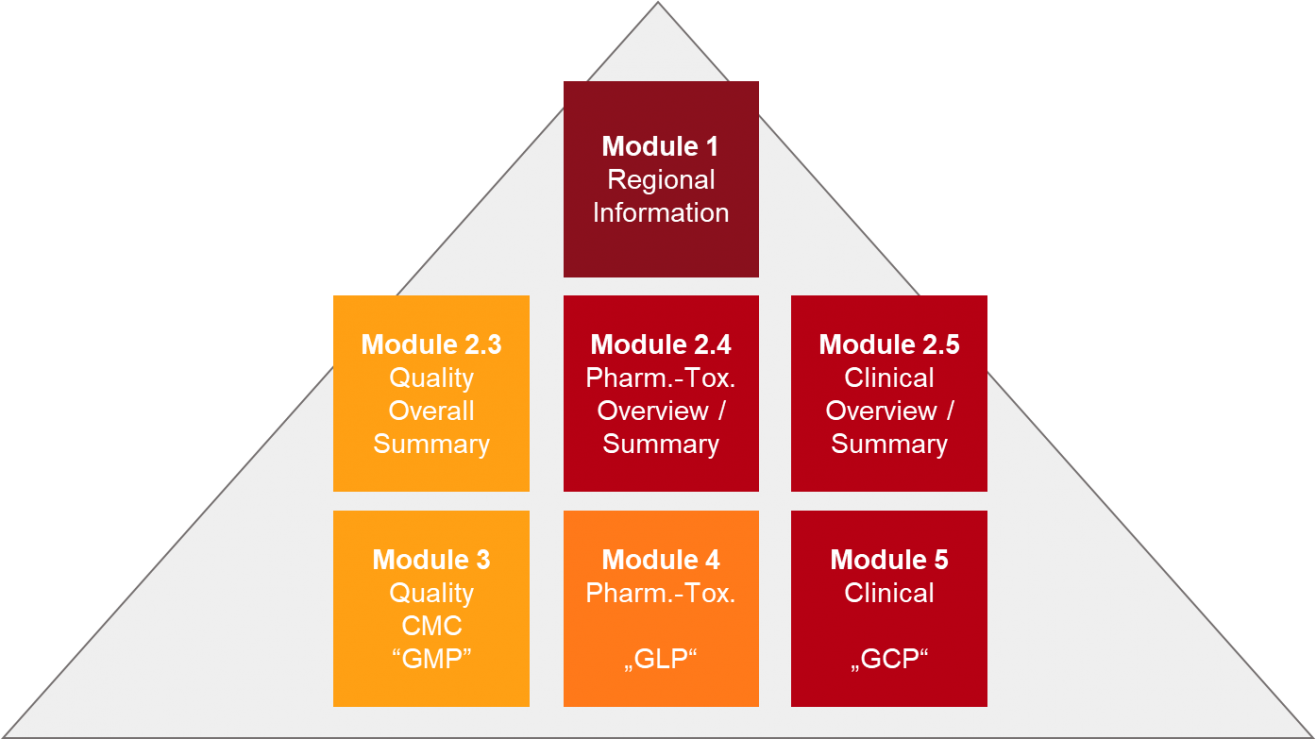
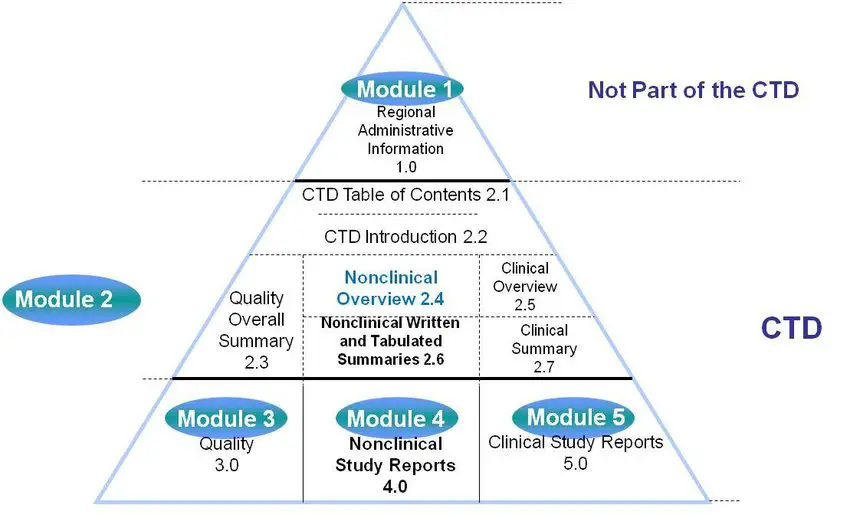
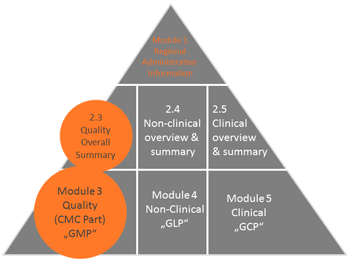
PDF) Preparation and Review of Chemistry, Manufacturing and Control (CMC) Sections of CTD Dossier for Marketing Authorization | Dhruvi Patel - Academia.edu

The Future of CMC Regulatory Submissions: Streamlining Activities Using Structured Content and Data Management - ScienceDirect

Transitioning Chemistry, Manufacturing, and Controls Content With a Structured Data Management Solution: Streamlining Regulatory Submissions - ScienceDirect

Architecture (left) and component size (right) of the CMC module. The... | Download Scientific Diagram