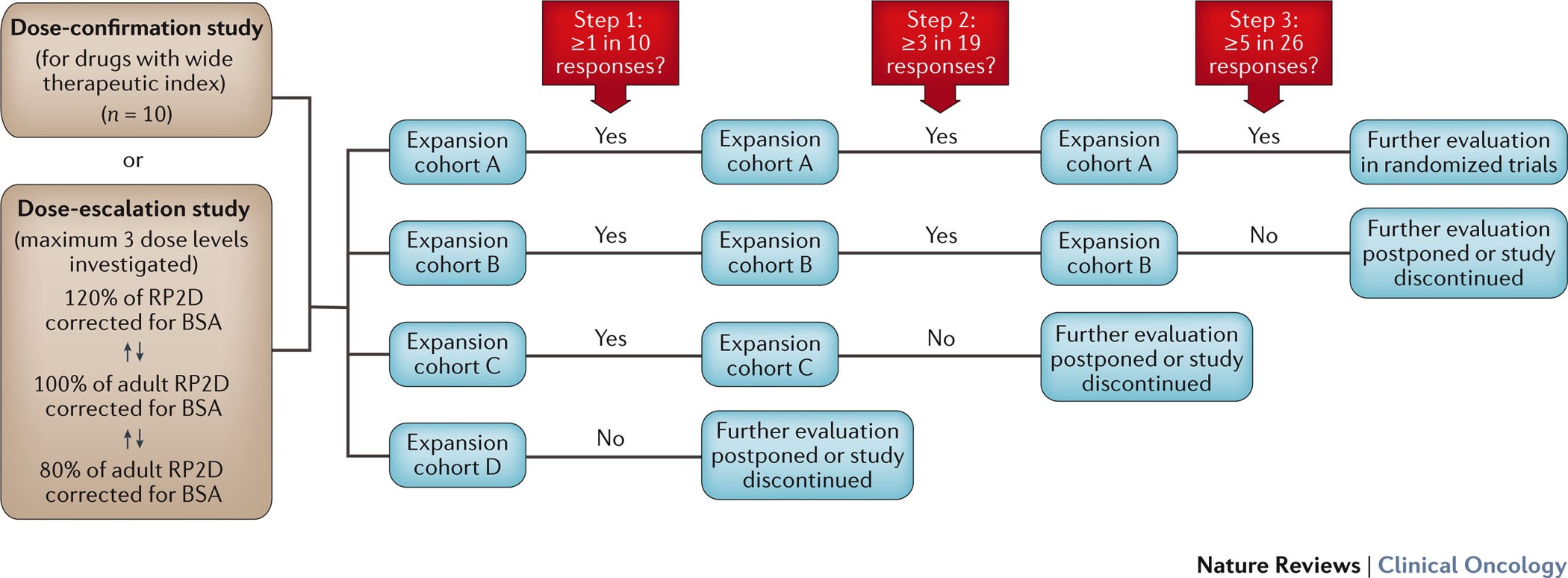
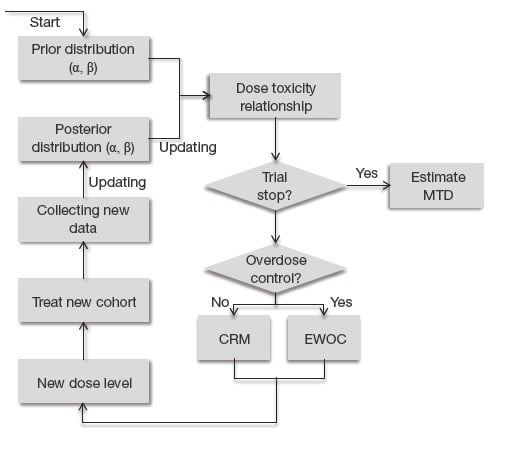
Genpro Research - Finding the right MTD/RP2D for phase 2 is the important goal of phase 1 clinical trials. The popular 3+3 design is being used widely to identify the correct dose,

A Bayesian Logistic Model with Covariate to Identify Optimal Dose for Heterogeneous Population in Phase I Oncology Trial Xin Wei, Mike Branson Celgene. - ppt download

Exposure driven dose escalation design with overdose control: Concept and first real life experience in an oncology phase I trial - ScienceDirect
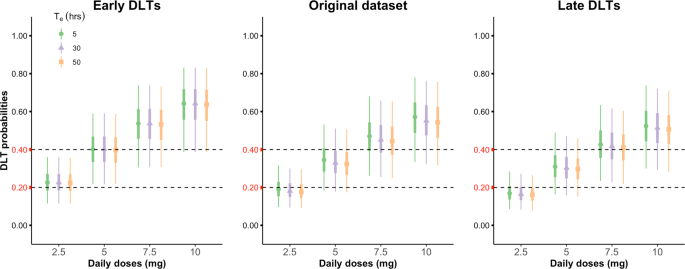
Phase I dose-escalation oncology trials with sequential multiple schedules | BMC Medical Research Methodology | Full Text
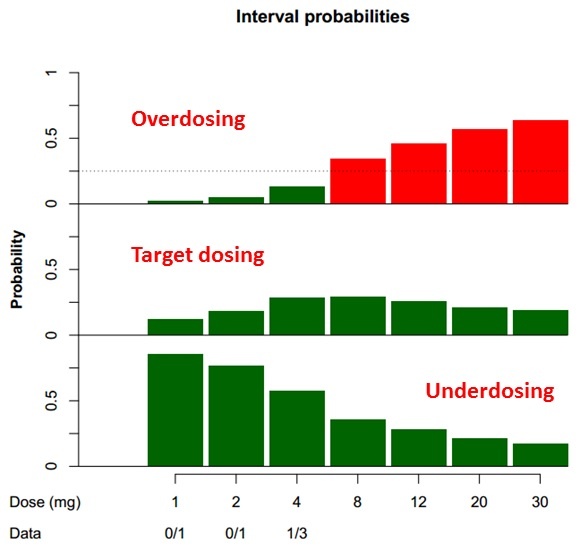
Bayesian designs in clinical trials | Data science, biostatistics, data transparency, bioinformatics and statistical programming

Contemporary dose-escalation methods for early phase studies in the immunotherapeutics era - ScienceDirect
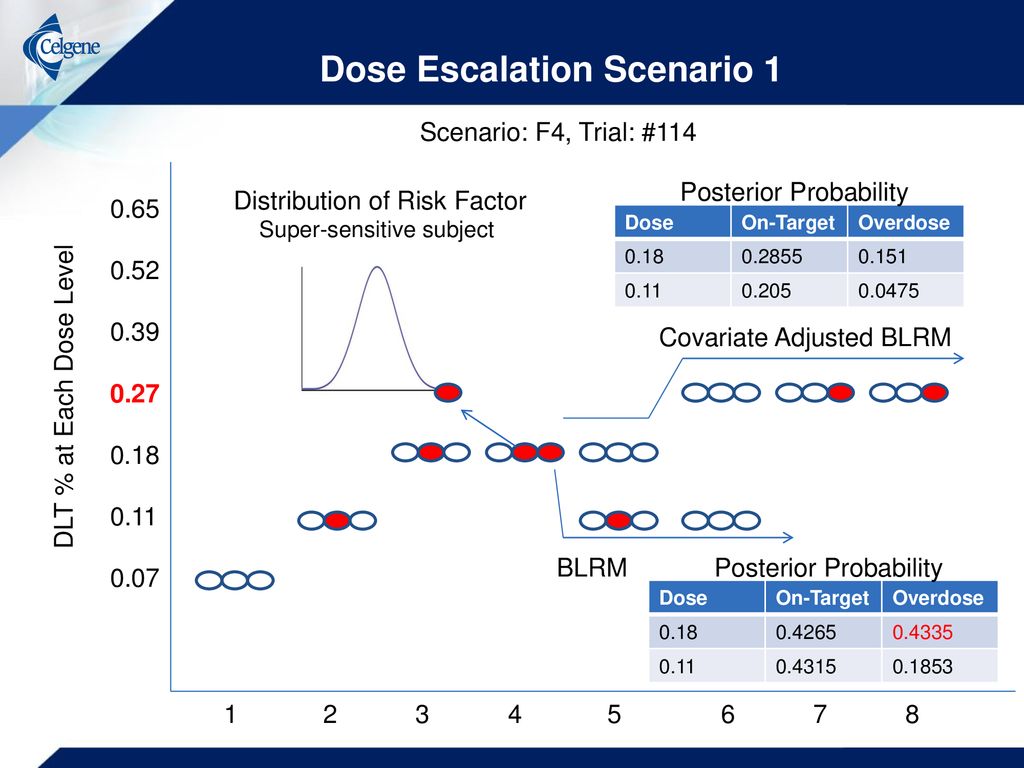
A Bayesian Logistic Model with Covariate to Identify Optimal Dose for Heterogeneous Population in Phase I Oncology Trial Xin Wei, Mike Branson Celgene. - ppt download

Improving the performance of Bayesian logistic regression model with overdose control in oncology dose‐finding studies - Zhang - 2022 - Statistics in Medicine - Wiley Online Library

A Bayesian Logistic Model with Covariate to Identify Optimal Dose for Heterogeneous Population in Phase I Oncology Trial Xin Wei, Mike Branson Celgene. - ppt download